17-10210
ProteoExtract® Native Cytoskeleton Enrichment
The ProteoExtract Native Cytoskeleton Enrichment & Staining Kit provides cytoskeleton purification detergent buffers that retain focal adhesion & actin-associated proteins while removing soluble cytoplasmic & nuclear proteins from the cell.
About This Item
Recommended Products
Quality Level
manufacturer/tradename
Chemicon®
ProteoExtract®
technique(s)
immunocytochemistry: suitable
immunofluorescence: suitable
application(s)
sample preparation
shipped in
dry ice
General description
http://www.nature.com/app_notes/nmeth/2012/121007/pdf/an8624.pdf
(Click Here!)
Rediscovering the Actin Cytoskeleton: New techniques leading to discoveries in cell migration, Dr. Richard Klemke, Morris Cancer Center, UCSD, USA.
The actin cytoskeleton is a highly dynamic network composed of actin polymers and a large variety of associated proteins. The functions of the actin cytoskeleton is to mediate a variety of essential biological functions in all eukaryotic cells, including intra- and extra-cellular movement and structural
Support (Chen, C.S., et al., 2003; Frixione E., 2000). To perform these functions, the organization of the actin cytoskeleton must be tightly regulated both temporally and spatially. Many proteins associated with the actin cytoskeleton are thus likely targets of signaling pathways controlling actin assembly. Actin cytoskeleton assembly is regulated at multiple levels, including the organization of actin monomers (G-actin) into actin polymers and the superorganization of actin polymers into a filamentous network (F-actin – the major constituent of microfilaments) (Bretscher, A., et al., 1994). This superorganization of actin polymers into a filamentous network is mediated by actin side-binding or cross-linking proteins (Dubreuil, R. R., 1991; Matsudaira, P., 1991; Matsudaira, P., 1994). The actin cytoskeleton is a dynamic structure that rapidly changes shape and organization in response to stimuli and cell cycle progression. Therefore, a disruption of its normal regulation may lead to cell transformations and cancer. Transformed cells have been shown to contain less F-actin than untransformed cells and exhibit atypical coordination of F-actin levels throughout the cell cycle (Rao, J.Y., et al., 1990). Orientational distribution of actin filaments within a cell is, therefore, an important determinant of cellular shape and motility.
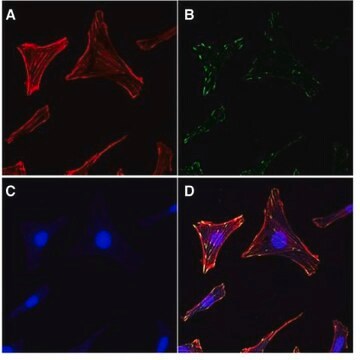
Focal adhesion and adherens junctions are membrane-associated complexes that serve as nucleation sites for actin filaments and as cross-linkers between the cell exterior, plasma membrane and actin cytoskeleton (Yamada, K.M., Geiger, B., 1997). The function of focal adhesions is structural, linking the ECM on the outside to the actin cytoskeleton on the inside. They are also sites of signal transduction, initiating signaling pathways in response to adhesion. Focal adhesions consist of integrin-type receptors that are attached to the extracellular matrix and are intracellularly associated with protein complexes containing vinculin (universal focal adhesion marker), talin, α-actinin, paxillin, tensin, zyxin and focal adhesion kinase (FAK) (Burridge, K., et al., 1990; Turner, C.E., Burridge, K., 1991).
Studying the proteins that associate with and regulate the actin cytoskeleton has been traditionally difficult because of the insolubility of the cytoskeleton in traditional detergents like Triton-X100. Work over the years has shown that many actin regulatory proteins/phospho-proteins upon activation move from the soluble cytoplasmic compartment to the insoluble actin cytoskeleton. The insolubility of these important proteins has made it difficult to study their biochemical changes, such as phosphorylation and nitrosylation, upon binding to actin. What is sorely needed is a cytoskeleton purification method that allows for the selective enrichment of cytoskeleton-associated proteins for detailed protein biochemical analyses. Such a method would provide the means to directly study this important pool of proteins in normal and diseased cytoskeletons.
Application
Cell Structure
Cytoskeleton
Packaging
Components
One bottle containing 3.5 mL of 20x Cytoskeleton Wash Buffer
One bottle containing 9 mL of ready to use Nuclear Extraction Buffer
One vial containing 100 µL of 1000x Protease Inhibitor Cocktail solution
One vial containing 150 µL of 1000x Sodium Orthovanadate solution
One vial containing 50 µL of 200x donkey anti-mouse FITC conjugate
One bottle containing 12.5 mL of 10x Blocking/Permeabilization Buffer
One vial containing 15 µg lyophilized TRITC-Conjugated Phalloidin
One vial containing 100 µL of 200x DAPI
One vial containing 100 µL of 100x anti-Vinculin mouse monoclonal IgG1 antibody
One vial containing 16 µL of 500x anti-GAPDH mouse monoclonal IgG1 antibody
Storage and Stability
Legal Information
Disclaimer
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Eye Dam. 1 - Flam. Liq. 2 - Skin Corr. 1C - Skin Sens. 1 - STOT SE 1
Target Organs
Eyes,Central nervous system
Storage Class Code
3 - Flammable liquids
Flash Point(F)
50.0 °F
Flash Point(C)
10 °C
Regulatory Listings
Regulatory Listings are mainly provided for chemical products. Only limited information can be provided here for non-chemical products. No entry means none of the components are listed. It is the user’s obligation to ensure the safe and legal use of the product.
FSL
Group 4: Flammable liquids
Type 1 petroleums
Hazardous rank II
Water insoluble liquid
JAN Code
17-10210:
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service