Key Documents
Safety Information
1.01695
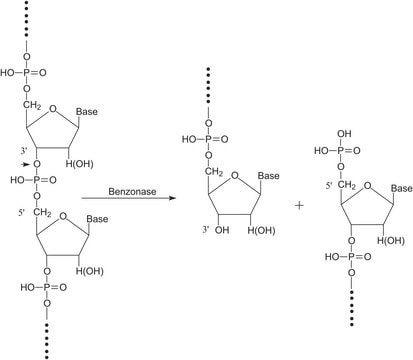
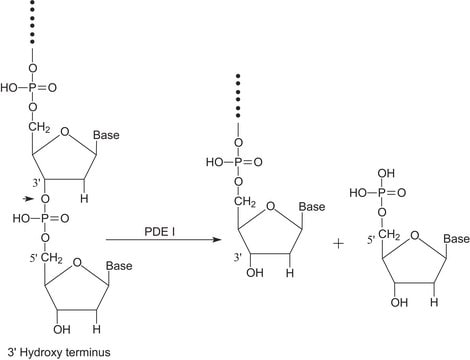
Benzonase®
suitable for biopharmaceutical production EMPROVE® bio
Pharma Manufacturing
Synonym(s):
Endonuclease from Serratia marcescens
About This Item
Recommended Products
biological source
Serratia marcescens
Quality Level
recombinant
expressed in E. coli
product line
EMPROVE® Bio
Assay
≥99% (SDS-PAGE)
form
buffered aqueous glycerol solution
concentration
≥250 units/μL
pH
8.0 (25 °C in H2O)
application(s)
pharma/biopharma processes
shipped in
dry ice
storage temp.
-10 to -25°C
Looking for similar products? Visit Product Comparison Guide
General description
Benzonase® endonuclease is the smart solution for DNA removal in viral vaccine and viral vector production and has proven its value for over 30 years. Balancing efficiency and regulatory compliance by delivering reliability and high-quality due manufacturing under good manufacturing practices (GMP ICH Q7). Our extensive documentation packages of Emprove® dossiers and the availability of a FDA Drug Master File type II, supports you in filing your vaccine manufacturing process to the authorities.
M-Clarity Program
As part of our EMPROVE® Program, our raw materials are offered with EMPROVE® Dossiers which provide comprehensive, up-to-date documentation to help you navigate regulatory challenges, manage risks, and improve your manufacturing processes.
Our comprehensive portfolio of downstream process chemicals not only provides biopharmaceutical manufacturers with high-quality raw materials for production of classical and novel therapies, but also helps them get to market faster and simplify regulatory challenges. Ranging from non-GMP grades for low-risk application, to IPEC-PQG GMP for higher-risk applications, we have products covering all your manufacturing needs.
Application
Biochem/physiol Actions
Legal Information
also commonly purchased with this product
related product
Storage Class Code
10 - Combustible liquids
WGK
WGK 1
Regulatory Listings
Regulatory Listings are mainly provided for chemical products. Only limited information can be provided here for non-chemical products. No entry means none of the components are listed. It is the user’s obligation to ensure the safe and legal use of the product.
JAN Code
1016958001:
1016950004:
MX11016950001:
1016950001:
1016950003:
1016950000:
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Related Content
A relatively templated manufacturing process can be used to produce live vector vaccines. Challenges exist, however, and include yield loss due to sterile filtration, vector aggregation and scaling adherent cultures. Learn all about this core platform technology and how expertise—empowered by collaboration— can overcome manufacturing challenges.
Manufacturing viral vaccines is complex and there is no templated approach. Each process must be tailored to the shape, size, nature, physico-chemical behavior, stability, and host specificity of the virus. Learn all about this core platform technology and how expertise—empowered by collaboration— can overcome manufacturing challenges.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service