695874
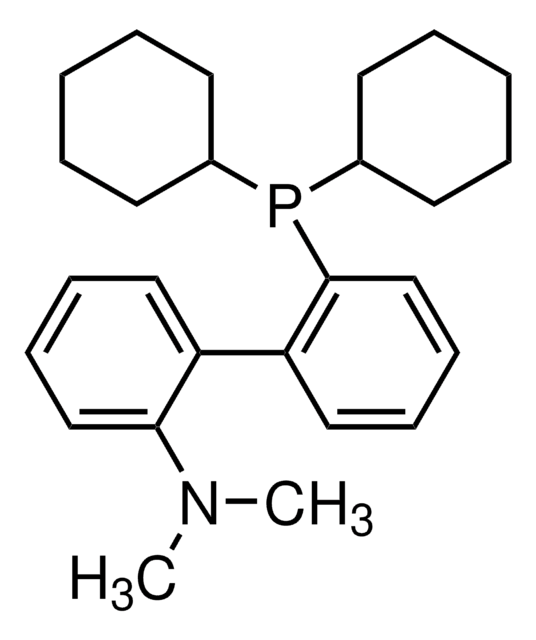
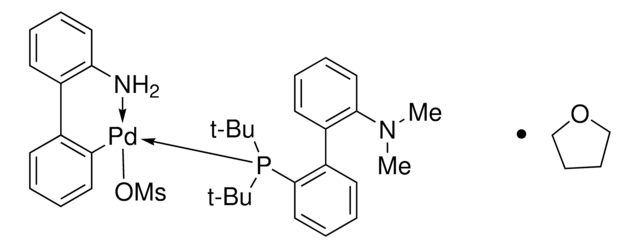
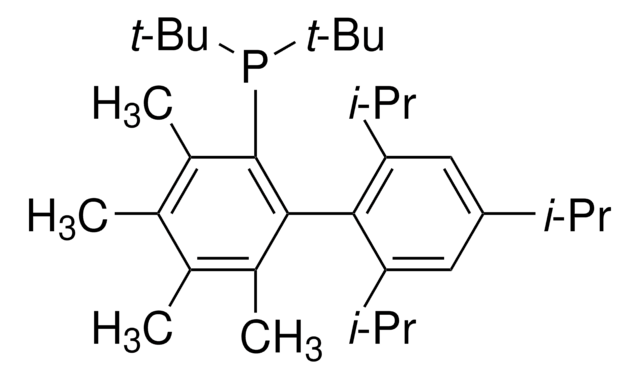
t-BuDavePhos
Synonym(s):
2′-(Di-tert-butylphosphino)-N,N-dimethylbiphenyl-2-amine, t-Butyl DavePhos, 2-Di-tert-butylphosphino-2′-(N,N-dimethylamino)biphenyl, tBuDavePhos
About This Item
Recommended Products
form
solid
Quality Level
reaction suitability
reagent type: ligand
reaction type: Arylations
reagent type: ligand
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reagent type: ligand
reaction type: C-X Bond Formation
mp
114-118 °C
functional group
phosphine
SMILES string
CN(C)c1ccccc1-c2ccccc2P(C(C)(C)C)C(C)(C)C
InChI
1S/C22H32NP/c1-21(2,3)24(22(4,5)6)20-16-12-10-14-18(20)17-13-9-11-15-19(17)23(7)8/h9-16H,1-8H3
InChI key
PHLPNEHPCYZBNZ-UHFFFAOYSA-N
Related Categories
Application
Dialkylbiaryl phosphine ligand often used for Pd-catalyzed reactions in a range of contexts.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Chronic 4 - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Regulatory Listings
Regulatory Listings are mainly provided for chemical products. Only limited information can be provided here for non-chemical products. No entry means none of the components are listed. It is the user’s obligation to ensure the safe and legal use of the product.
JAN Code
695874-VAR:
695874-BULK:
695874-5G:
695874-1G:
695874-250MG:
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Buchwald phosphine ligands for C-C, C-N, and C-O bond formation.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service