N3538
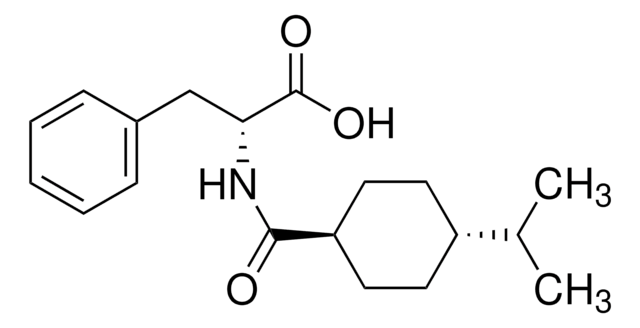
Nateglinide
≥98% (HPLC), solid
Sinonimo/i:
Fastic, N-[(trans-4-isopropylcyclohexyl)carbonyl]-D-phenylalanine, Starlix, Starsis
About This Item
Prodotti consigliati
Livello qualitativo
Saggio
≥98% (HPLC)
Stato
solid
Colore
white to off-white
Solubilità
DMSO: >5 mg/mL
H2O: insoluble
Ideatore
Novartis
Temperatura di conservazione
room temp
Stringa SMILE
CC(C)[C@@H]1CC[C@H](CC1)C(=O)N[C@H](Cc2ccccc2)C(O)=O
InChI
1S/C19H27NO3/c1-13(2)15-8-10-16(11-9-15)18(21)20-17(19(22)23)12-14-6-4-3-5-7-14/h3-7,13,15-17H,8-12H2,1-2H3,(H,20,21)(H,22,23)/t15-,16-,17-/m1/s1
OELFLUMRDSZNSF-BRWVUGGUSA-N
Informazioni sul gene
human ... ABCC8(6833) , KCNJ11(3767)
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Azioni biochim/fisiol
Caratteristiche e vantaggi
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Certificati d'analisi (COA)
Non trovi la versione di tuo interesse?
Se hai bisogno di una versione specifica, puoi cercare il certificato tramite il numero di lotto.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Articoli
Sigma-Aldrich offers many products related to potassium channels for your research needs.
Glucose metabolism is regulated by the opposing actions of insulin and glucagon. Insulin is released from pancreatic ß cells in response to high blood glucose levels and regulates glucose metabolism through its actions on muscle, liver, and adipose tissue.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.