C4520
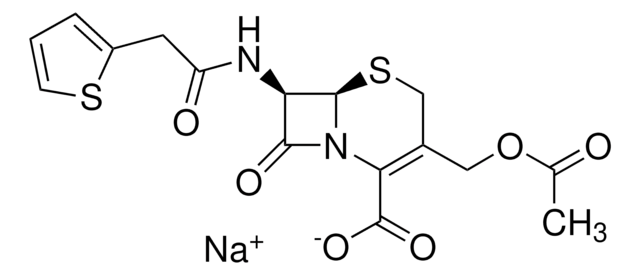
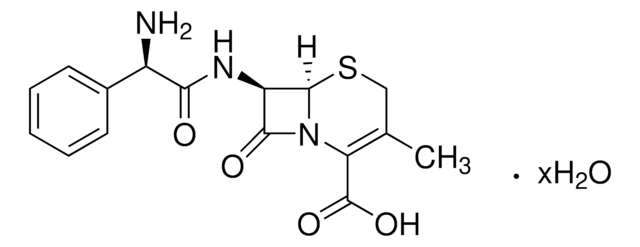
Cephalothin sodium salt
Sinonimo/i:
7-(2-Thienylacetamido)cephalosporanic acid sodium salt, Cephalotin sodium salt
About This Item
Prodotti consigliati
Stato
crystalline powder
Livello qualitativo
Spettro attività antibiotica
Gram-negative bacteria
Gram-positive bacteria
Modalità d’azione
cell wall synthesis | interferes
Temperatura di conservazione
2-8°C
Stringa SMILE
[Na+].CC(=O)OCC1=C(N2[C@H](SC1)[C@H](NC(=O)Cc3cccs3)C2=O)C([O-])=O
InChI
1S/C16H16N2O6S2.Na/c1-8(19)24-6-9-7-26-15-12(14(21)18(15)13(9)16(22)23)17-11(20)5-10-3-2-4-25-10;/h2-4,12,15H,5-7H2,1H3,(H,17,20)(H,22,23);/q;+1/p-1/t12-,15-;/m1./s1
VUFGUVLLDPOSBC-XRZFDKQNSA-M
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
Applicazioni
Azioni biochim/fisiol
Antimicrobial spectrum: Effective aginast both Gram-positive and Gram-negative bacteria.
Mode of Resistance: Production of cephalosporinase will inactivate the product.
Altre note
Avvertenze
Danger
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Resp. Sens. 1 - Skin Sens. 1
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 2
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.