358673
Tetrabutylammonium fluoride on silica gel
~1.5 mmol/g (F-)
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
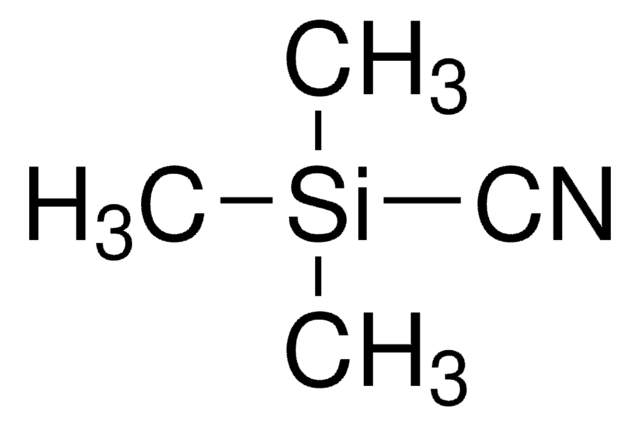
Formula condensata:
(CH3CH2CH2CH2)4N(F)
Peso molecolare:
261.46
Beilstein:
3570522
Numero CE:
Numero MDL:
Codice UNSPSC:
12352200
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Capacità
~1.5 mmol/g (F-)
Stringa SMILE
[F-].CCCC[N+](CCCC)(CCCC)CCCC
InChI
1S/C16H36N.FH/c1-5-9-13-17(14-10-6-2,15-11-7-3)16-12-8-4;/h5-16H2,1-4H3;1H/q+1;/p-1
FPGGTKZVZWFYPV-UHFFFAOYSA-M
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Acute Tox. 4 Oral - Aquatic Chronic 3 - Eye Irrit. 2 - Repr. 2 - Skin Irrit. 2
Rischi supp
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Mehrdad Balandeh et al.
Journal of the Electrochemical Society, 164(9), G99-G103 (2017-09-12)
Electrochemical fluorination of methyl(phenylthio)acetate was achieved using tetrabutylammonium fluoride (TBAF). Electrochemical fluorination was performed under potentiostatic anodic oxidation using an undivided cell in acetonitrile containing TBAF and triflic acid. The influence of several parameters including: oxidation potential, time, temperature, sonication
Santosh Kumar Behera et al.
Photochemical & photobiological sciences : Official journal of the European Photochemistry Association and the European Society for Photobiology, 14(12), 2225-2237 (2015-10-31)
The mechanism for the dual emission of 2-(4'-N,N-dimethylaminophenyl)imidazo[4,5-c]pyridine (DMAPIP-c) in protic solvents was investigated by synthesizing and studying its analogues. Theoretical calculations were carried out to corroborate the experimental findings. The deprotonation studies suggest that the enhancement in the TICT
Delia López-Velázquez et al.
Carbohydrate polymers, 125, 224-231 (2015-04-11)
We have synthesized and characterized five members of a homologous series of side chain polymers of hydroxypropyl cellulose esters obtained by homogeneous esterification with 6-[4'-(ethoxycarbonyl)biphenyl-4-yloxy]hexanoic acid. Two acylation procedures were studied. One procedure involved the acid chloride derivative and the
Eric P Gillis et al.
Journal of medicinal chemistry, 58(21), 8315-8359 (2015-07-23)
The role of fluorine in drug design and development is expanding rapidly as we learn more about the unique properties associated with this unusual element and how to deploy it with greater sophistication. The judicious introduction of fluorine into a
Michelle L Ingalsbe et al.
Bioorganic & medicinal chemistry letters, 19(17), 4984-4987 (2009-08-08)
Due to the increasing number of strains of drug-resistant bacteria, the development of new antibiotics has become increasingly important. The antibacterial properties of quaternary amines and their derivatives on both Gram-positive and Gram-negative bacteria are well known. However, an encompassing
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.