COHISR-RO
Roche
cOmplete™ His-Tag Purification Resin
suspension, suitable for protein purification, matrix Sepharose-CL 6B
About This Item
Recommended Products
form
suspension
packaging
pkg of 200 mL (05893801001 [settled resin volume])
pkg of 25 mL (05893682001 [settled resin volume])
manufacturer/tradename
Roche
technique(s)
protein purification: suitable
matrix
Sepharose-CL 6B
storage temp.
2-8°C
Related Categories
General description
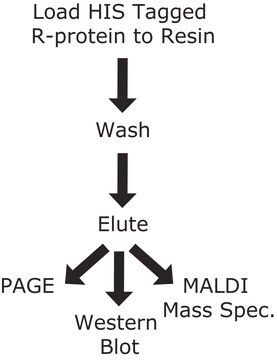
Binding capacity: ≥ 40mg protein per ml bed volume of resin. The binding capacity of the resin to various types of proteins may vary according to the protein characteristics such as the size of the protein. cOmplete His-Tag Purification Columns bind with a high specificity to the polyhistidine-tagged protein. As a consequence, the binding kinetics may appear to be different when compared to conventional metal chelate matrices. Full capacity of cOmplete His-Tag Purification Columns can be achieved by allowing more time for the protein to bind to the resin by lowering the flow rate during the chromatography purification procedure.
Maximal linear flow rate: 1,420cm/hour
Recommended volumetric flow rate: The volumetric flow rate is a function of the cross section of the column. Using the following formula, a linear flow rate can be converted to a volumetric flow (mL/min.): Linear flow rate (cm/hour) × column cross sectional area (cm2)/60. The column cross sectional area is defined as Π× r2, whereas Π is the constant pi and r is the inner radius of the column.
Recommended imidazole concentration for load/wash: Nonspecific binding of proteins without a His-tag is low. Use up to 5mM imidazole in load and/or wash buffers. If establishing a new assay for purification of His-tag proteins with cOmplete His-Tag Purification Columns, do not use imidazole. If, e.g., the purity of the His-tag protein needs to be improved following this first step, use imidazole in a final concentration of up to 5mM in a second step.
Recommended imidazole concentration for elution: Up to 500 mM. Please note: Contrary to other available resins, bound His-tagged protein typically elutes from cOmplete His-Tag Purification Columns with a lower imidazole concentration, e.g., 25 to 45mM.
Compatibility for long term storage: 20% ethanol, pH 4.0 to pH 9.0
Compatibility during chromatography: The resin is compatible with 10mM EDTA, 10mM DTT during the purification (1 hour incubation), 6M guanidinium-HCl, 8M urea, pH 2.0 to pH 14.0.
Compatibility during cleaning: 4% SDS
In addition, the resin maintains its binding capacity whether you are using low- or high-molecular weight proteins, and its specificity allows a one-step purification. By using one of the strongest chelators available, minimal contamination of your flow-through fractions with metal ions is ensured, safeguarding your downstream applications.
The cOmplete His-Tag Purification Resin is also available as prepacked format: with 1mL or 5mL resin.
Application
Purifying a protein of interest is often essential for determining its function, structure, or interactions, for raising specific antibodies, or preparing enzymes for practical applications. Isolation of naturally expressed proteins from their original source can be a complex process involving numerous chromatographic steps. Recombinant protein expression in dedicated host organisms can greatly simplify this task. Such expression systems generally ensure higher expression levels. Fusing the target protein to a tag also confers advantageous binding ability to an affinity matrix.
Protein Purification using Immobilized Ni2+
The most common technique in affinity purification of proteins involves engineering a sequence of 6 to 14 histidines into the N- or C-terminal (or even on an exposed loop) of the protein. Such polyhistidine stretches bind strongly to divalent metal ions such as nickel and cobalt. This effect can be used to separate proteins. Metal ions can be immobilized on a matrix using a chelator, which still allows the ion to interfere with the polyhistidine tag of the protein. When these his-tagged proteins are passed through a column containing immobilized metal ions, the proteins bind via the tag to the column. Nearly all untagged proteins pass through the column. The protein is released from the column by elution with either imidazole, which competes with the polyhistidine tag for binding to the column, or by a decrease in pH, which decreases the affinity of the tag for the resin. While this procedure is generally used for the purification of recombinant proteins with an engineered affinity tag, it can also be used for natural proteins with an inherent affinity for divalent cations.
His-Tags
Ideally, the His-tagged target protein binds much stronger to the Ni2+ chelate matrix than endogenous histidine-containing protein of the expression host. Relative binding strength depends on how many histidines can bind simultaneously to the matrix (avidity effect). Longer His-tags confer stronger binding and better separation of the target from potentially contaminating host proteins. The classic His-tag has six consecutive histidines. Tags with 10 to 14 histidines may produce a better purification. Most importantly, His-tagged proteins can be purified using Ni2+ chelate matrices under both native and denaturing conditions. Due to their hydrophilic and flexible nature, these matrices increase the solubility of the target proteins and only rarely interfere with protein function. This unique combination of features enables the His-tag to be a versatile tool for a wide range of protein purification applications.
Features and Benefits
- Use the buffer conditions best suited to your protein: Keep your protein comfortable and let it, not your purification resin, determine whether you use DTT, EDTA, or other buffer substances.
- Repeatedly obtain highly pure protein: Single step purification without resin recharging.
- Protect your protein from toxic nickel: Reduce protein oxidation and aggregation caused by resins that leach nickel.
- Work in a safe and eco-friendly environment: Avoid handling of toxic nickel and completely eliminate disposal costs.
Physical form
Other Notes
Legal Information
Signal Word
Warning-Warning
Hazard Statements
Hazard Classifications
Flam. Liq. 3
Storage Class Code
3 - Flammable liquids
WGK
WGK 1
Flash Point(F)
93.2 °F
Flash Point(C)
34 °C
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Get maximum protection after protein isolation by using protease and phosphatase inhibitor cocktail tablets from Roche.
Protocols
cOmplete™ His-Tag Purification Resin Protocol & Troubleshooting
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service