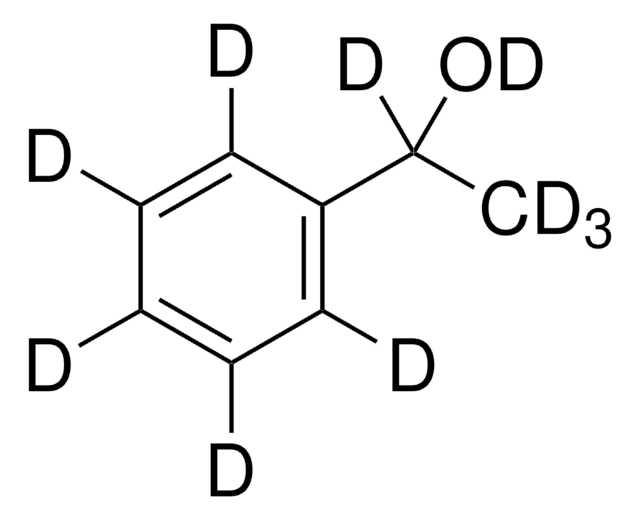
P13800
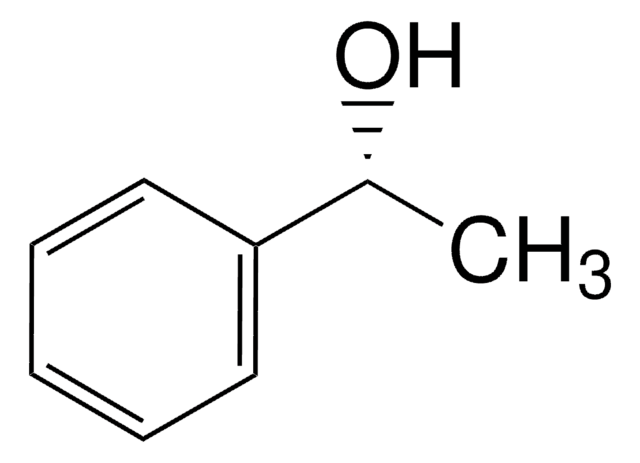
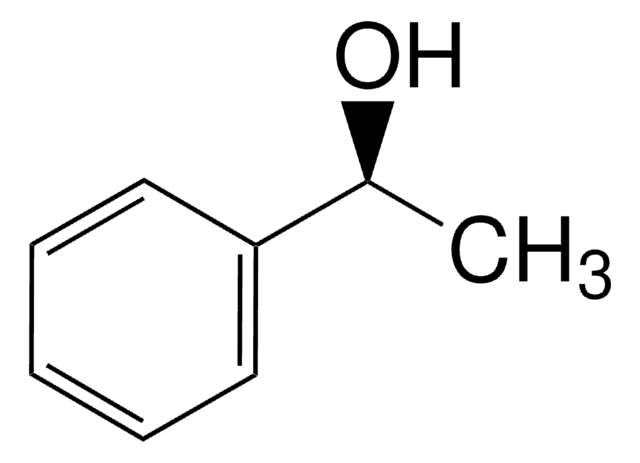
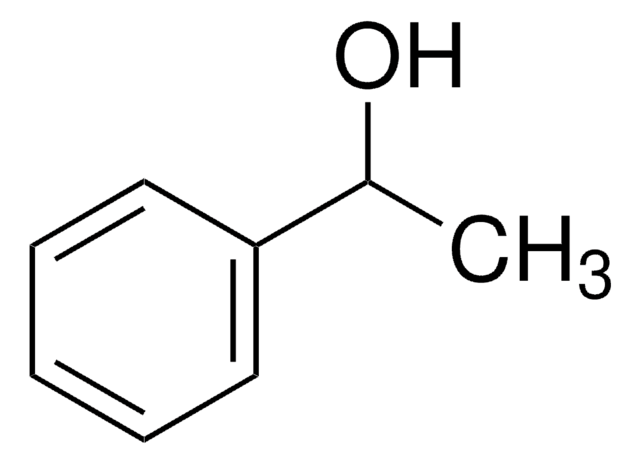
1-Phenylethanol
98%
Synonym(s):
(±)-1-Phenylethanol, (±)-α-Methylbenzyl alcohol, Methyl phenyl carbinol, Styrallyl alcohol, Styrene alcohol
About This Item
Recommended Products
vapor density
4.21 (vs air)
Quality Level
vapor pressure
0.1 mmHg ( 20 °C)
Assay
98%
refractive index
n20/D 1.527 (lit.)
bp
204 °C/745 mmHg (lit.)
mp
19-20 °C (lit.)
density
1.012 g/mL at 25 °C (lit.)
SMILES string
CC(O)c1ccccc1
InChI
1S/C8H10O/c1-7(9)8-5-3-2-4-6-8/h2-7,9H,1H3
InChI key
WAPNOHKVXSQRPX-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- 1-Phenylethanol can be used in the acid catalyzed Ritter reaction to synthesize various N-benzylacetamides.
- It is one of the key precursors for the synthesis of [60] fullerene-fused isochroman derivatives via palladium-catalyzed hydroxyl-directed cyclization.
- It is used in Friedlander synthesis of quinolines where 1-phenylethanol reacts with 2-aminobenzylic alcohol derivatives to yield corresponding quinones.
- It can also be used in the regioselective, iridium-catalyzed multicomponent synthesis of pyrimidines.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 1
Flash Point(F)
187.9 °F - Pensky-Martens closed cup
Flash Point(C)
86.6 °C - Pensky-Martens closed cup
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Global Trade Item Number
| SKU | GTIN |
|---|---|
| P13800-1KG | 4061834354642 |
| P13800-25G | 4061834354710 |
| P13800-500G | 4061834354727 |
| P13800-5KG |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service