665290
(S,S,S)-(+)-(3,5-Dioxa-4-phosphacyclohepta[2,1-a:3,4-a′]dinaphthalen-4-yl)bis(1-phenylethyl)amine
97%
Synonym(s):
(+)-N,N-Bis[(1S)-1-phenylethyl]- dinaphtho[2,1-d:1′,2′-f][1,3,2]dioxaphosphepin- 4-amine, (11bR)
About This Item
Recommended Products
Quality Level
Assay
97%
form
solid
mp
88-89 °C
functional group
amine
phenyl
SMILES string
C[C@H](N([C@@H](C)c1ccccc1)P2Oc3ccc4ccccc4c3-c5c(O2)ccc6ccccc56)c7ccccc7
InChI
1S/C36H30NO2P/c1-25(27-13-5-3-6-14-27)37(26(2)28-15-7-4-8-16-28)40-38-33-23-21-29-17-9-11-19-31(29)35(33)36-32-20-12-10-18-30(32)22-24-34(36)39-40/h3-26H,1-2H3/t25-,26-/m0/s1
InChI key
LKZPDRCMCSBQFN-UIOOFZCWSA-N
Application
- Iridium-catalyzed allylic etherification of acyclic, achiral allylic carbonates with potassium silanolates to form chiral allylic alcohols.
- Palladium-catalyzed asymmetric allylic cyclisation of N-tosyl and N-benzyl carbonates to form the corresponding pyrrolidine and piperidine derivatives, respectively.
- Intramolecular iridium-catalyzed allylic cyclizationof (E)-allylic methyl carbonates to form 2,5-trans/cis pyrrolidine derivatives.
Features and Benefits
- Superior enantiocontrol in numerous transformations
- High activities at low catalyst loadings
- Hydrogenations under low-pressure conditions
Legal Information
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
A diverse array of these chiral, monodentate phosphoramidites based on the privileged BINOL platform. The MonoPhos™ family has exhibited high levels of enantiocontrol in synthetic transformations ranging from metal-catalyzed asymmetric 1,4-additions of organometallic reagents to allylic alkylations to desymmetrization of meso-cycloalkene oxides.
In collaboration with DSM, we are pleased to offer a range of MonoPhos™ ligands for the research market.† Feringa and co-workers have invented a diverse array of these chiral, monodentate phosphoramidites based on the privileged BINOL platform.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service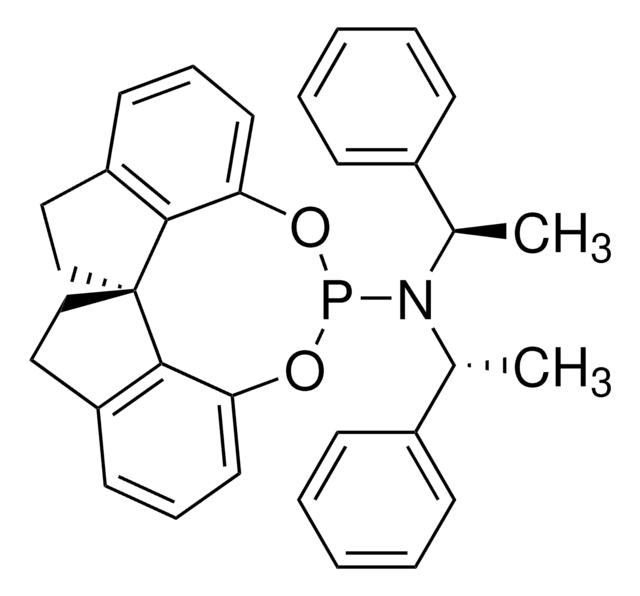
![(S,R,R)-(+)-(3,5-Dioxa-4-phosphacyclohepta[2,1-a:3,4-a′]dinaphthalen-4-yl)bis(1-phenylethyl)amine 95% (HPLC)](/deepweb/assets/sigmaaldrich/product/structures/366/790/7555ef31-5d0b-45c9-ad40-5dfd0fe85125/640/7555ef31-5d0b-45c9-ad40-5dfd0fe85125.png)
![(S)-(+)-N-(3,5-Dioxa-4-phosphacyclohepta[2,1-a;3,4-a′]dinaphthalen-4-yl)-dibenzo[b,f]azepine ≥95% (elemental analysis)](/deepweb/assets/sigmaaldrich/product/structures/575/489/d54360f9-5a59-43f2-bc44-42f5fa92b588/640/d54360f9-5a59-43f2-bc44-42f5fa92b588.png)
![(S)-(+)-(3,5-Dioxa-4-phosphacyclohepta[2,1-a;3,4- a′]dinaphthalen-4-yl)dimethylamine 97%](/deepweb/assets/sigmaaldrich/product/structures/400/008/628143de-3954-440a-ba9c-4c0ff8e44663/640/628143de-3954-440a-ba9c-4c0ff8e44663.png)
![1,8-Diazabicyclo[5.4.0]undec-7-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)
![4,7,13,16,21,24-Hexaoxa-1,10-diazabicyclo[8.8.8]hexacosane 98%](/deepweb/assets/sigmaaldrich/product/structures/189/812/8a6555e5-8de6-4236-865f-19339cee3634/640/8a6555e5-8de6-4236-865f-19339cee3634.png)

![(S,R)-(+)-(3,5-Dioxa-4-phosphacyclohepta[2,1-a:3,4-a′]dinaphthalen-4-yl)-(1-phenylethyl)amine 96%](/deepweb/assets/sigmaaldrich/product/structures/340/157/5071e653-a834-4559-9aa7-4eb2d3774e42/640/5071e653-a834-4559-9aa7-4eb2d3774e42.png)
![(−)-Bis[(S)-1-phenylethyl]amine 99%](/deepweb/assets/sigmaaldrich/product/structures/336/455/d6f04f0e-9bcc-4d67-a94d-d153e39209e1/640/d6f04f0e-9bcc-4d67-a94d-d153e39209e1.png)