270083
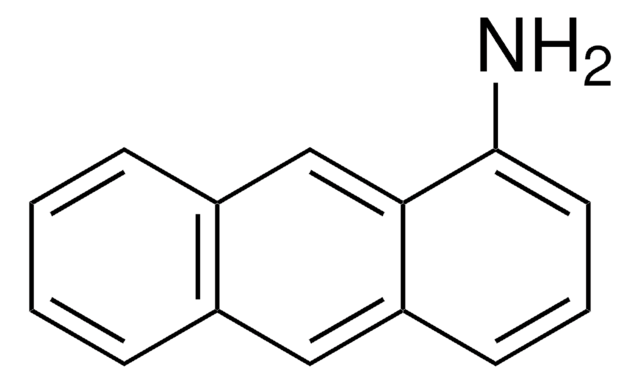
9-(Methylaminomethyl)anthracene
99%
Synonym(s):
(Anthracen-9-ylmethyl)-N-methylamine, 1-Anthracen-9-yl-N-methylmethanamine, 9-(N-Methylaminomethyl)anthracene, N-Methyl-9-anthracenemethanamine
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C16H15N
CAS Number:
Molecular Weight:
221.30
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
99%
form
solid
mp
57-59 °C (lit.)
functional group
amine
storage temp.
2-8°C
SMILES string
CNCc1c2ccccc2cc3ccccc13
InChI
1S/C16H15N/c1-17-11-16-14-8-4-2-6-12(14)10-13-7-3-5-9-15(13)16/h2-10,17H,11H2,1H3
InChI key
WRVHTDZJMNUGQN-UHFFFAOYSA-N
Related Categories
General description
9-(Methylaminomethyl)anthracene is an isocyanate derivatizing reagent.
Application
9-(Methylaminomethyl)anthracene (MAMA) was used as a model drug in the study of the effect of enzymatic degradation on the release behaviors of MAMA by UV-Vis spectroscopy. It was also used as a reagent in a sensitive method for determination of isocyanates in air by HPLC via UV or fluorescence detection.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Journal of Liquid Chromatography, 3, 971-971 (1980)
X Y Xiong et al.
Journal of controlled release : official journal of the Controlled Release Society, 108(2-3), 263-270 (2005-10-04)
Poly(lactic acid) (PLA) was successfully grafted to both ends of Pluronic F127 block copolymer (PEO-PPO-PEO) to obtain amphiphilic PLA-F127-PLA block copolymers. The effect of enzymatic degradation on the release behaviors of hydrophobic model drug 9-(methylaminomethyl)anthracene (MAMA) from PLA-F127-PLA nano-particles with
R P Streicher et al.
American Industrial Hygiene Association journal, 57(10), 905-913 (1996-10-01)
Analytical reference standards generally are not available for non-monomeric isocyanate species, making accurate identification and quantitation by high-performance liquid chromatography (HPLC) difficult. A successful derivatizing reagent must react rapidly with all isocyanate groups, the derivatized isocyanate must be detectable selectively
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service