T9935
Trypsin from bovine pancreas
essentially salt-free, lyophilized powder, ≥9,000 BAEE units/mg protein, BioReagent, suitable for cell culture
Synonym(s):
Serine Protease 1
About This Item
Recommended Products
product line
BioReagent
Quality Level
form
essentially salt-free, lyophilized powder
specific activity
≥9,000 BAEE units/mg protein
mol wt
23.8 kDa
composition
protein, ≥80%
technique(s)
cell culture | mammalian: suitable
solubility
hydrochloric acid: soluble 1 mM
storage temp.
−20°C
Looking for similar products? Visit Product Comparison Guide
Application
Biochem/physiol Actions
Serine protease inhibitors, including DFP, TLCK, APMSF, AEBSEF, and aprotinin, amongst others, will inhibit Trypsin.
Components
Caution
Unit Definition
Preparation Note
For applications that involve EDTA, solubilizing trypsin should be done with a buffered salt solution containing no Ca2+ or Mg2+.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Resp. Sens. 1 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Protocols
This procedure is for products with a specification for Trypsin activity using Na-Benzoyl-L-arginine ethyl ester (BAEE) as a substrate. The procedure is a continuous spectrophotometric rate determination (A253, Light path = 1 cm).
Related Content
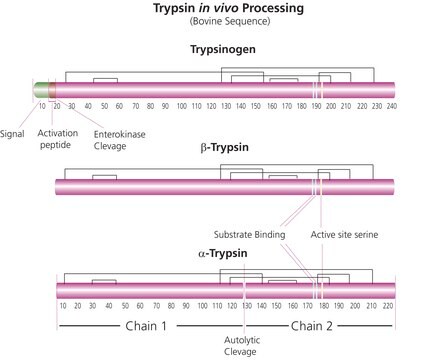
Trypsin is an enzyme in the serine protease class that consists of a polypeptide chain of 223 amino acid residues. Multiple sources, grades and formulations of trypsin specifically designed for research applications are available.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service