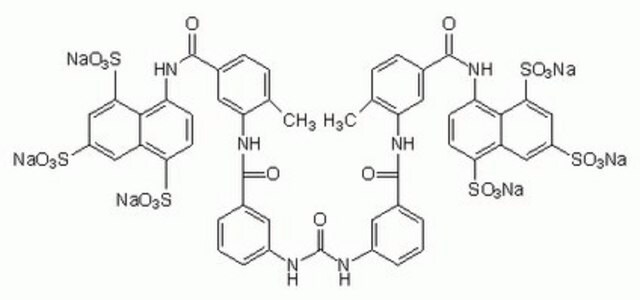
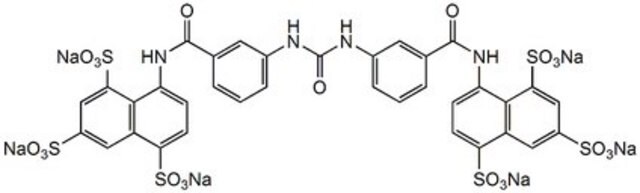
S2671
Suramin sodium salt
≥98% (TLC)
Synonym(s):
Suramine sodium salt
About This Item
Recommended Products
Quality Level
Assay
≥98% (TLC)
form
powder
impurities
<15% water (Karl Fischer)
color
white to light brown
solubility
H2O: >10 mg/mL
SMILES string
[Na+].[Na+].[Na+].[Na+].[Na+].[Na+].Cc1ccc(cc1NC(=O)c2cccc(NC(=O)Nc3cccc(c3)C(=O)Nc4cc(ccc4C)C(=O)Nc5ccc(c6cc(cc(c56)S([O-])(=O)=O)S([O-])(=O)=O)S([O-])(=O)=O)c2)C(=O)Nc7ccc(c8cc(cc(c78)S([O-])(=O)=O)S([O-])(=O)=O)S([O-])(=O)=O
InChI
1S/C51H40N6O23S6.6Na/c1-25-9-11-29(49(60)54-37-13-15-41(83(69,70)71)35-21-33(81(63,64)65)23-43(45(35)37)85(75,76)77)19-39(25)56-47(58)27-5-3-7-31(17-27)52-51(62)53-32-8-4-6-28(18-32)48(59)57-40-20-30(12-10-26(40)2)50(61)55-38-14-16-42(84(72,73)74)36-22-34(82(66,67)68)24-44(46(36)38)86(78,79)80;;;;;;/h3-24H,1-2H3,(H,54,60)(H,55,61)(H,56,58)(H,57,59)(H2,52,53,62)(H,63,64,65)(H,66,67,68)(H,69,70,71)(H,72,73,74)(H,75,76,77)(H,78,79,80);;;;;;/q;6*+1/p-6
InChI key
VAPNKLKDKUDFHK-UHFFFAOYSA-H
Gene Information
human ... EGF(1950) , FGF1(2246) , P2RX1(5023) , P2RY1(5028) , PDGFA(5154) , PLD1(5337)
Looking for similar products? Visit Product Comparison Guide
General description
Application
- as a potent blocker of rat P2Y2 purinergic receptor
- to study its effects on short circuit current (Isc) across strial marginal cells
- to show its effects on initial burst of glial activity
- to assess its neuroprotective potential in rats
- as a purinoceptor inhibitor, to explore the mechanism of action of extracellular adenosine triphosphate (eATP)
Biochem/physiol Actions
Other Notes
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
EGF Signaling: Tracing Cancer's Path
Epigenetic modifications are thought to occur through two key interconnected processes—DNA methylation and the covalent modification of histones.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service