G6637
β-Galactose Dehydrogenase from Pseudomonas fluorescens
recombinant, expressed in E. coli, ammonium sulfate suspension, ≥50 units/mg protein (biuret)
Synonym(s):
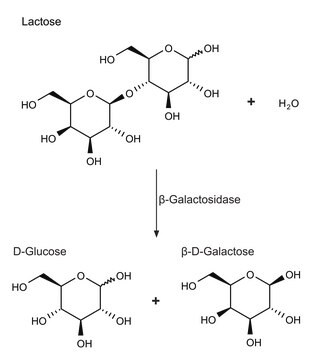
D-Galactose:NAD+ 1-oxidoreductase
About This Item
Recommended Products
recombinant
expressed in E. coli
Quality Level
form
ammonium sulfate suspension
specific activity
≥50 units/mg protein (biuret)
shipped in
wet ice
storage temp.
2-8°C
Looking for similar products? Visit Product Comparison Guide
Application
Biochem/physiol Actions
Unit Definition
Physical form
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Instructions for working with enzymes supplied as ammonium sulfate suspensions
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service