33820
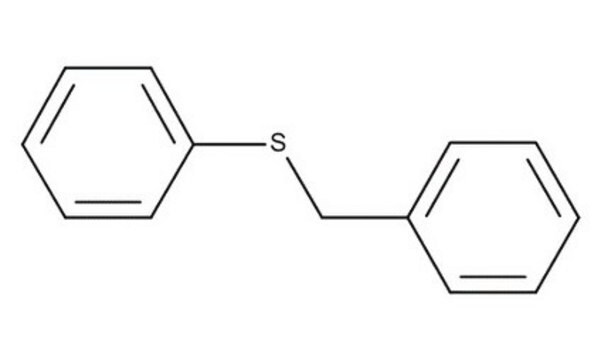
Dibenzyl sulfide
≥95.0% (HPLC)
Synonym(s):
Benzyl sulfide
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
(C6H5CH2)2S
CAS Number:
Molecular Weight:
214.33
Beilstein:
1911157
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
≥95.0% (HPLC)
form
solid
mp
44-47 °C (lit.)
functional group
phenyl
thioether
SMILES string
C(SCc1ccccc1)c2ccccc2
InChI
1S/C14H14S/c1-3-7-13(8-4-1)11-15-12-14-9-5-2-6-10-14/h1-10H,11-12H2
InChI key
LUFPJJNWMYZRQE-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Sulfur K-edge XANES spectra of dibenzyl sulfide has been measured. Adsorption modes of dibenzyl disulphide on bare and iron-coated silver in 0.5M HCl was studied by surface-enhanced Raman scattering (SERS) spectroscopy. Dibenzyl sulfide undergoes oxidation to dibenzyl sulfoxide by SiO2-HNO3/KBr.
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Vered Heleg-Shabtai et al.
Advances in experimental medicine and biology, 733, 53-61 (2011-11-22)
The improvements in Raman instrumentation have led to the development of -portable, simple to operate, Raman instruments that can be used for on-site analysis of substances relevant for homeland security purposes such as chemical and biological warfare and explosives materials.Raman
Eun Kyoung Ryu et al.
Bioorganic & medicinal chemistry, 12(5), 859-864 (2004-02-26)
Benzyl 4-halobenzyl and ally benzyl disulfide were synthesized as diallyl disulfide analogues and their tumor growth inhibitory effects on the cancer cells (SNU C5 and MCF-7) were comparable to that of diallyl disulfide, indicating that the disulfide functional group was
Robert Beck et al.
Dalton transactions (Cambridge, England : 2003), (37)(37), 4981-4983 (2008-09-20)
C-H activation through the coordination of a benzyl sulfide anchoring group with a C-S bond cleavage at a Co(v) center constitutes a regiospecific access to four- and five-membered metallacycles under mild conditions.
Chemo and homoselective catalytic oxidation of sulfides to sulfoxides with supported nitric acid on silica gel and poly vinyl pyrrolidone (PVP) catalyzed by KBr and/or NaBr.
Zolfigol MA, et al.
Catalysis Communications, 9(8), 1739-1744 (2008)
B Lei et al.
Journal of bacteriology, 178(19), 5699-5705 (1996-10-01)
The oxidation of dibenzothiophene to dibenzothiophene sulfone has been linked to the enzyme encoded by the sox/dszC gene from Rhodococcus sp. strain IGTS8 (S. A. Denome, C. Oldfield, L. J. Nash, and K. D. Young, J. Bacteriol. 176:6707-6717, 1994; C.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service