261432
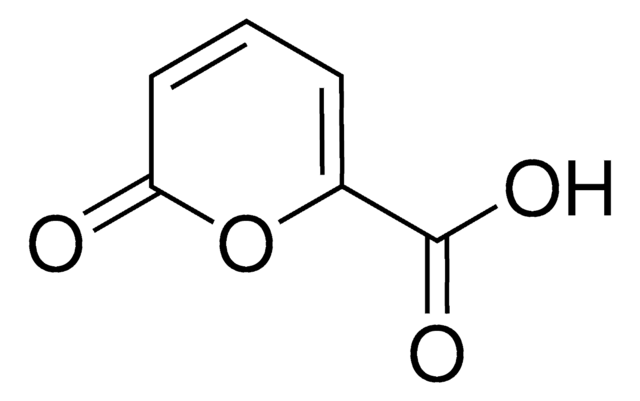
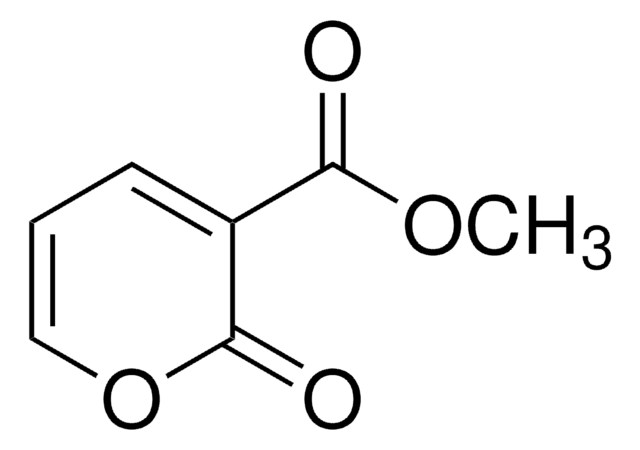
Methyl coumalate
98%
Synonym(s):
Methyl 2-oxo-2H-pyran-5-carboxylate, Methyl 2-pyrone-5-carboxylate
About This Item
Recommended Products
Assay
98%
form
solid
bp
178-180 °C/60 mmHg (lit.)
mp
65-67 °C (lit.)
functional group
ester
SMILES string
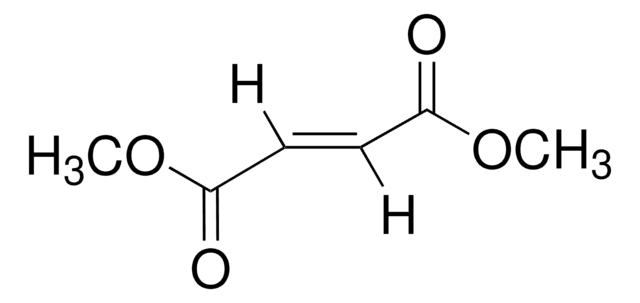
COC(=O)C1=COC(=O)C=C1
InChI
1S/C7H6O4/c1-10-7(9)5-2-3-6(8)11-4-5/h2-4H,1H3
InChI key
HHWWWZQYHPFCBY-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
The Diels–Alder reaction is the reaction between a conjugated diene and an alkene (dienophile) to form unsaturated six-membered rings. Since the reaction involves the formation of a cyclic product via a cyclic transition state, it is also referred to as a "cycloaddition".
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service