195464
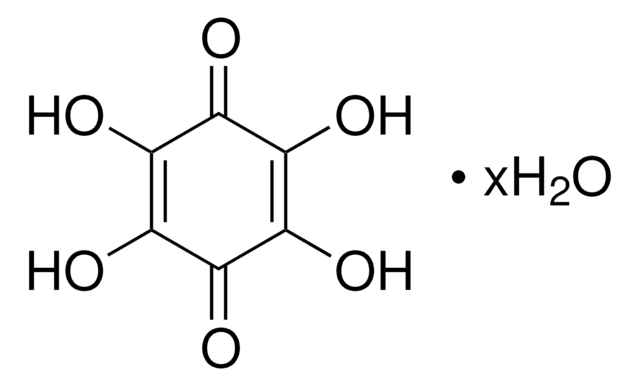
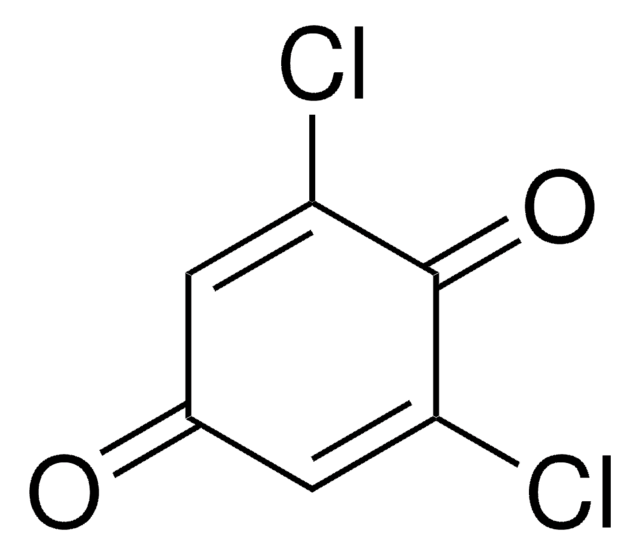
2,5-Dihydroxy-1,4-benzoquinone
98%
Synonym(s):
2,5-Dihydroxy-2,5-cyclohexadiene-1,4-dione, 2,5-Dihydroxy-p -benzoquinone, 2,5-Dihydroxycyclohexa-2,5-diene-1,4-dione, Anilic acid
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Linear Formula:
(HO)2C6H2(=O)2
CAS Number:
Molecular Weight:
140.09
EC Number:
MDL number:
UNSPSC Code:
12162002
PubChem Substance ID:
NACRES:
NA.23
Recommended Products
Assay
98%
mp
235 °C (dec.) (lit.)
SMILES string
OC1=CC(=O)C(O)=CC1=O
InChI
1S/C6H4O4/c7-3-1-4(8)6(10)2-5(3)9/h1-2,7,10H
InChI key
QFSYADJLNBHAKO-UHFFFAOYSA-N
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Jan Bohuslavek et al.
Biodegradation, 16(4), 353-362 (2005-05-04)
Hydroxyquinol, a common metabolite of aromatic compounds, is readily auto-oxidized to hydroxyquinone. Enzymatic activities that metabolized hydroxyquinone were observed from the cell extracts of Sphingobium chlorophenolicum ATCC 39723. An enzyme capable of transforming hydroxyquinone was partially purified, and its activities
M Ahmed et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 56(5), 965-981 (2000-05-16)
The electronic absorption spectra of 1,4-benzoquinone (BQ) and its 2,5-dihydroxy and tetrahydroxy derivatives have been studied in detail. The interpretation of the electronic bands is made on the basis of PPP and CNDO calculations. It is found that the pi
P G Jene et al.
Acta crystallographica. Section C, Crystal structure communications, 57(Pt 6), 730-734 (2001-06-16)
The X-ray structure of 1,2,4,5-tetrahydroxybenzene (benzene-1,2,4,5-tetrol) monohydrate, C6H6O4*H2O, (I), reveals columns of 1,2,4,5-tetrahydroxybenzene parallel to the b axis that are separated by 3.364 (12) and 3.453 (11) A. Molecules in adjacent columns are tilted relative to each other by 27.78
Y Sun et al.
Guang pu xue yu guang pu fen xi = Guang pu, 20(6), 790-792 (2003-08-27)
In this paper, we report the Ab initio calculation and experimental investigation of the vibrational frequencies of 4,5-dihydroxy-1,2-benzoquinone and 2,5-dihydroxy-1,4-benzoquinone. The main IR and Raman absorption bands of them have been assigned, learned from other strong points to offset one
A Pawlukojć et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 59(3), 537-542 (2003-01-14)
Inelastic neutron scattering (INS) spectra of solid 2,5-dihydroxy-1,4-benzoquinone were measured and compared with IR and Raman data. The INS spectrum is very well reproduced in the region below 1000 cm(-1) by DFT calculations on the B3LYP/6-311++G** level using Gaussian and
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service