All Photos(2)
About This Item
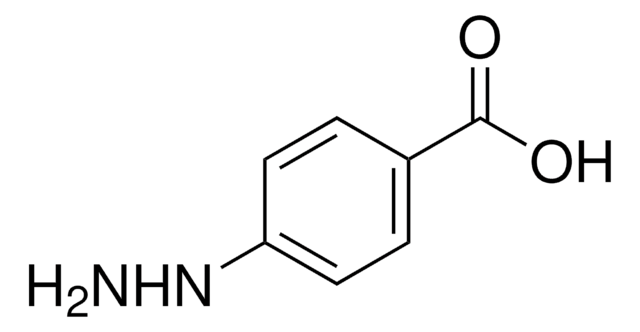
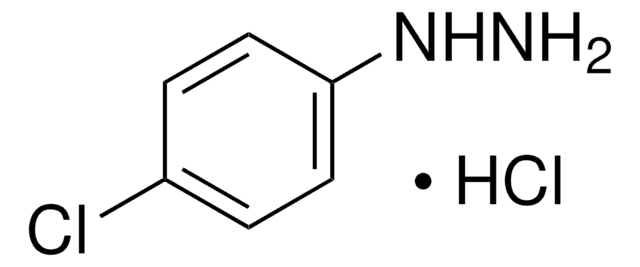
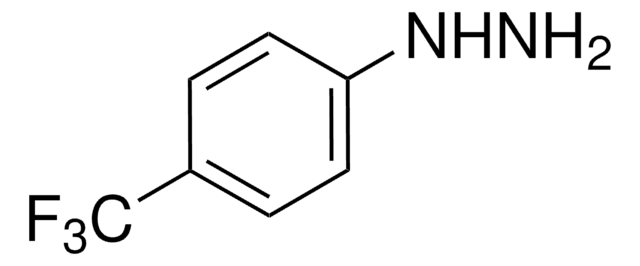
Linear Formula:
H2NNHC6H4CO2H · HCl
CAS Number:
Molecular Weight:
188.61
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
mp
253 °C (dec.) (lit.)
functional group
carboxylic acid
hydrazine
storage temp.
2-8°C
SMILES string
Cl.NNc1ccc(cc1)C(O)=O
InChI
1S/C7H8N2O2.ClH/c8-9-6-3-1-5(2-4-6)7(10)11;/h1-4,9H,8H2,(H,10,11);1H
InChI key
XHLQMKQBCHYRLC-UHFFFAOYSA-N
General description
4-Hydrazinobenzoic acid (p-Hydrazinobenzoic acid, HBA) has been reported as an ingredient of the cultivated mushroom Agaricus bisporus. It is carcinogenic to rodents. Mechanism of DNA damage by 4-hydrazinobenzoic acid has been studied by using 32P-labeled DNA fragments obtained from the human p53 and p16 tumor suppressor genes. It may be used for the preparation of 4-(3-(2-(tert-butoxycarbonylamino)ethyl)-5-hydroxy-1H-pyrazol-1-yl)benzoic acid.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Synthesis of 3-(2-aminoethyl)-5-hydroxy-1H-pyrazole derivatives.
Groselj U, et al.
ARKIVOC (Gainesville, FL, United States), 3, 49-65 (2012)
B M McManus et al.
Laboratory investigation; a journal of technical methods and pathology, 57(1), 78-85 (1987-07-01)
p-Hydrazinobenzoic acid (HBA), an ingredient of the cultivated mushroom Agaricus bisporus, was given in hydrochloride form at a dosage of 0.125% in drinking water for life to randomly bred Swiss mice. Previous studies had demonstrated that either synthetic or naturally
Yun-Gon Kim et al.
Chemistry & biology, 15(3), 215-223 (2008-03-22)
Glycan recognition leading to cell-cell interactions, signaling, and immune responses is mediated by various glycan-binding proteins (GBPs) showing highly diverse ligand specificities. We describe here a rapid glycan immobilization technique via 4-hydrazinobenzoic acid (HBA)-functionalized beads and its application to high-throughput
Yan-Juan Gu et al.
International journal of nanomedicine, 6, 2889-2898 (2011-12-02)
Single-walled carbon nanotubes (SWNTs) have been identified as an efficient drug carrier. Here a controlled drug-delivery system based on SWNTs coated with doxorubicin (DOX) through hydrazone bonds was developed, because the hydrazone bond is more sensitive to tumor microenvironments than
X Wang et al.
Carbohydrate research, 332(2), 191-196 (2001-07-04)
p-Hydrazinobenzenesulfonic acid was explored as an ultraviolet labeling reagent for capillary electrophoresis of mono-, di- and trisaccharides. The labeling reaction that produces p-sulfophenylhydrazines took less than 8 min, and introduced both chromphore and charged groups into the carbohydrate molecules. The
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service