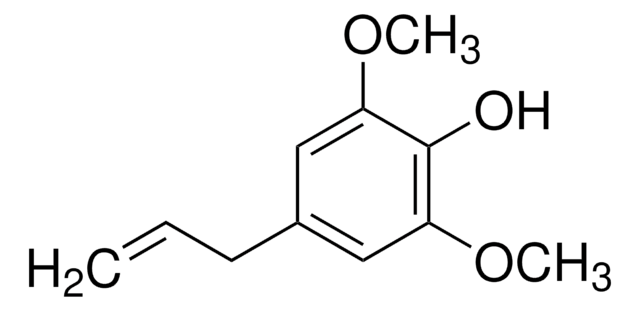
404586
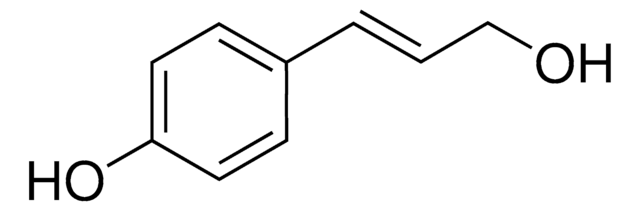
Sinapyl alcohol
technical grade, 80%
Synonym(s):
4-Hydroxy-3,5-dimethoxycinnamyl alcohol
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
HOC6H2(OCH3)2CH=CHCH2OH
CAS Number:
Molecular Weight:
210.23
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
grade
technical grade
Quality Level
Assay
80%
form
solid
mp
61-65 °C (lit.)
functional group
hydroxyl
storage temp.
2-8°C
SMILES string
COc1cc(\C=C\CO)cc(OC)c1O
InChI
1S/C11H14O4/c1-14-9-6-8(4-3-5-12)7-10(15-2)11(9)13/h3-4,6-7,12-13H,5H2,1-2H3/b4-3+
InChI key
LZFOPEXOUVTGJS-ONEGZZNKSA-N
General description
Sinapyl alcohol, a monolignol, is a primary lignin monomer. It has been evaluated for anti-inflammatory and antinociceptive activities. It participates in the initial stages in the biosynthesis of lignin. Coupling reactions of sinapyl alcohol and sinapyl p-hydroxybenzoate has been reported. Preparation of sinapyl alcohol by selective 1,2-reduction of corresponding cinnamate esters using diisobutylaluminium hydride as reducing agent has been studied.
Application
Sinapyl alcohol may be employed in the preparation of lignin, a highly stable biopolymer.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Fachuang Lu et al.
Organic & biomolecular chemistry, 2(20), 2888-2890 (2004-10-14)
Cross-coupling of sinapyl p-hydroxybenzoate and sinapyl alcohol produces an 8-8-cross-coupled product that is also detected in lignifying poplar tissues, implicating sinapyl p-hydroxybenzoate as a lignin precursor.
Zhen Fang et al.
Bioresource technology, 99(9), 3424-3430 (2007-09-21)
Decomposition of organosolve lignin in water/phenol solutions was studied in a 50 nL micro-reactor coupled with optical, Raman and infrared microscopies at temperatures up to 600 degrees C and water densities up to 1165 kg/m3. It was found that when
Tomoaki Kawabata et al.
Biomolecules, 10(7) (2020-07-19)
The pits of Japanese apricot, Prunus mume Sieb. et Zucc., which are composed of stones, husks, kernels, and seeds, are unused by-products of the processing industry in Japan. The processing of Japanese apricot fruits generates huge amounts of waste pits
Jun Shigeto et al.
Journal of plant research, 130(1), 203-210 (2016-11-27)
Most of the known 4-coumarate:coenzyme A ligase (4CL) isoforms lack CoA-ligation activity for sinapic acid. Therefore, there is some doubt as to whether sinapic acid contributes to sinapyl alcohol biosynthesis. In this study, we characterized the enzyme activity of a
A possible mechanism for the oxidation of sinapyl alcohol by peroxidase-dependent reactions in the apoplast: enhancement of the oxidation by hydroxycinnamic acids and components of the apoplast.
Takahama U, et al.
Plant Physiology, 37(4), 499-504 (1996)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service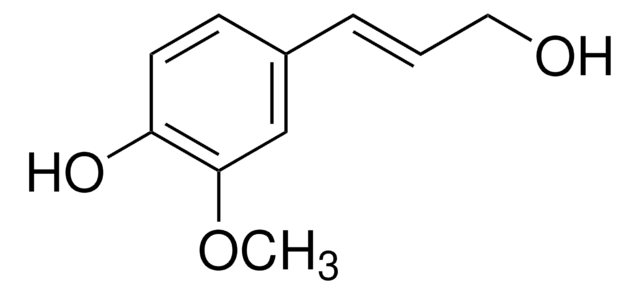





![4-[2-(4-Allyl-2,6-dimethoxyphenoxy)-1-hydroxypropyl]-2-methoxyphenol ≥95% (LC/MS-ELSD)](/deepweb/assets/sigmaaldrich/product/structures/103/717/7c015be4-f4aa-4720-b290-f5eaaaae5de4/640/7c015be4-f4aa-4720-b290-f5eaaaae5de4.png)