About This Item
Recommended Products
Quality Level
Assay
≥95%
reaction suitability
reagent type: reductant
refractive index
n20/D 1.397 (lit.)
bp
95 °C (lit.)
mp
−93 °C (lit.)
density
0.677 g/mL at 25 °C (lit.)
SMILES string
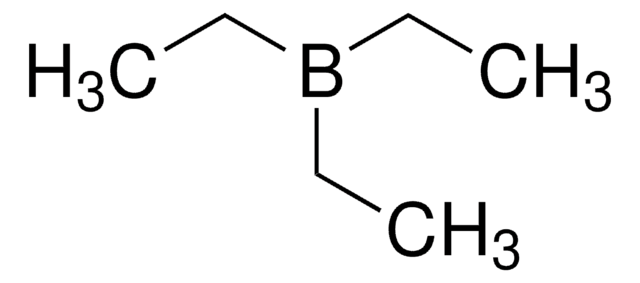
CCB(CC)CC
InChI
1S/C6H15B/c1-4-7(5-2)6-3/h4-6H2,1-3H3
InChI key
LALRXNPLTWZJIJ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- Enantioselective umpolung allylation of aldehydes
- Preparation of tetramethylammonium trialkylphenylborate salts
Catalyst for:
- Radical reductions of alkyl bromides and iodides bearing electron withdrawing groups with N-heterocyclic carbene boranes
- Synthesis of 1-substituted pyrrolines by N-diallylation of amines and ring-closing metathesis
- Decarboxylative C-C bond cleavage reactions
- Alkene hydrogenations
- Aminyl radical cyclizations onto silyl enol ethers
Modifier for single-site organochromium ethylene polymerization catalysts
Packaging
Compatible with the following:
- Aldrich® Sure/Pac™ station for liquefied gases Z566446
- PTFE Sealing tape Z104388 or Z221880
- Straight septum-inlet adapter Z118141 with septa Z565687 or Z565695
Legal Information
also commonly purchased with this product
recommended
septum inlet adapter
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Oral - Eye Dam. 1 - Pyr. Liq. 1 - Skin Corr. 1A
Storage Class Code
4.2 - Pyrophoric and self-heating hazardous materials
WGK
WGK 1
Personal Protective Equipment
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
We carry a large variety of electrophiles and nucleophiles that are widely used in C–C bond-forming reactions. This group of products contains many organometallic reagents as well as commonly-used alkylating and acylating reagents.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service