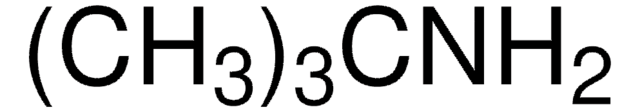All Photos(1)
About This Item
Linear Formula:
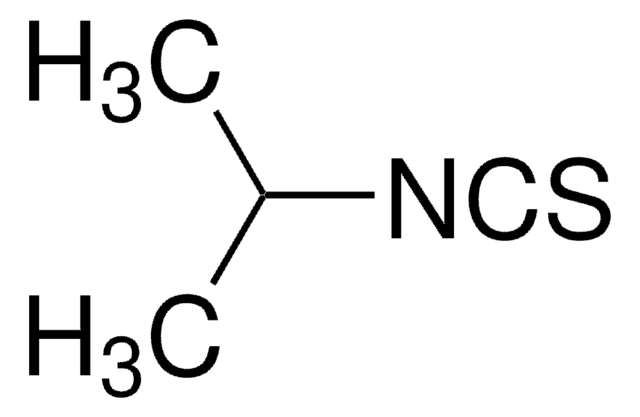
(CH3)3CNCS
CAS Number:
Molecular Weight:
115.20
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
99%
form
liquid
refractive index
n20/D 1.48 (lit.)
bp
30-32 °C/10 mmHg (lit.)
mp
10.5-11.5 °C (lit.)
density
0.908 g/mL at 25 °C (lit.)
functional group
amine
isothiocyanate
SMILES string
CC(C)(C)N=C=S
InChI
1S/C5H9NS/c1-5(2,3)6-4-7/h1-3H3
InChI key
ZFWFRTVIIMTOLY-UHFFFAOYSA-N
General description
Adsorption of tert-butyl isothiocyanate at the Ge (100) surface was studied using multiple internal reflection FTIR spectroscopy, X-ray photoelectron spectroscopy and density functional theory modeling.
Application
tert-Butyl isothiocyanate has been used to modify the pore size of top layer of heterostructured surface mounted metal-organic frameworks.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Eye Irrit. 2 - Flam. Liq. 3 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
100.4 °F - closed cup
Flash Point(C)
38 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Reaction of tert -butyl isocyanate and tert-butyl isothiocyanate at the Ge (100)- 2? 1 Surface.
Loscutoff PW, et al.
Surface Science, 604(19), 1791-1799 (2010)
Min Tu et al.
Dalton transactions (Cambridge, England : 2003), 42(45), 16029-16035 (2013-08-10)
Heterostructured surface mounted metal-organic frameworks (SURMOFs) [Cu2(NH2-bdc)2(dabco)] (B) on top of [Cu2(bdc)2(dabco)] (A) were deposited on pyridyl-terminated Au covered QCM substrate using a step-by-step liquid phase epitaxial growth method. Sequentially, the pore size of the top layer [Cu2(NH2-bdc)2(dabco)] (B) was
E Fontana et al.
Current drug metabolism, 6(5), 413-454 (2005-10-27)
The inhibition of human cytochrome P450s (CYPs) is one of the most common mechanisms which can lead to drug-drug interactions. The inhibition of CYPs can be reversible (competitive or non-competitive) or irreversible. Irreversible inhibition usually derives from activation of a
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service