179825
Borane dimethyl sulfide complex
Synonym(s):
(Dimethyl sulfide)trihydroboron, BMS, Borane-dimethyl sulfide
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
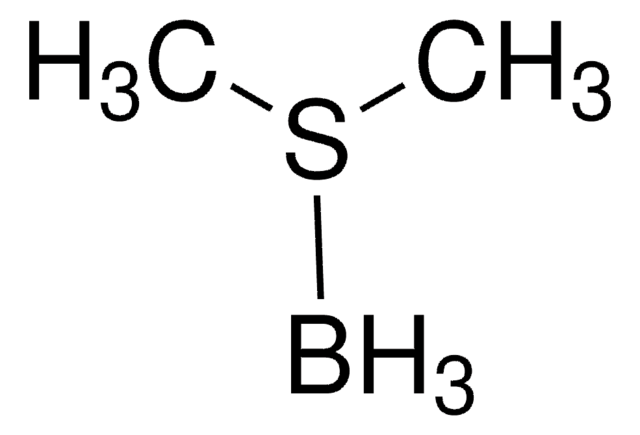
Linear Formula:
(CH3)2S · BH3
CAS Number:
Molecular Weight:
75.97
Beilstein:
3663489
EC Number:
MDL number:
UNSPSC Code:
12352112
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
form
liquid
Quality Level
reaction suitability
reagent type: reductant
density
0.801 g/mL at 25 °C (lit.)
functional group
thioether
storage temp.
2-8°C
SMILES string
B.CSC
InChI
1S/C2H6S.BH3/c1-3-2;/h1-2H3;1H3
InChI key
RMHDLBZYPISZOI-UHFFFAOYSA-N
Related Categories
General description
Borane dimethyl sulfide complex (BMS) is a commonly used reagent and a mediator for hydroboration reaction for the preparation of organoborane compounds, which are utilized as key intermediates in organic synthesis. BMS is also employed as a reducing agent for the reduction of various functional groups such as aldehydes, ketones, epoxides, esters, and carboxylic acids to corresponding alcohols.
Application
Borane-dimethyl sulfide (BH3 Me2S) can be used as a reagent:
- For the selective synthesis of 1,3,5-oxygenated compounds from dimethyl 3-oxoglutarate.
- For the conversion of ozonides to alcohols.
- In the CBS-catalyzed asymmetric reduction of ferrocenyl-1,3-diketones to 1,3-diols.
- For enantioselective reduction of ketones to chiral secondary alcohols in the presence of C3-symmetric tripodal hydroxyamide as a ligand.
- For the hydroboration reduction and other applications.
- With a dendrimeric supported L-pyrrolidinol in the asymmetric reduction of indanones and tetralones.
Packaging
The 25 mL Sure/Seal™ bottle is recommended as a single-use bottle. Repeated punctures will likely result in decreased performance of product.
Other Notes
10.0-10.2 M as BH3. May contain excess methyl sulfide.
Legal Information
Sure/Seal is a trademark of Sigma-Aldrich Co. LLC
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 3 Oral - Aquatic Chronic 3 - Eye Dam. 1 - Flam. Liq. 2 - Repr. 1B - Water-react 1
Storage Class Code
4.3 - Hazardous materials which set free flammable gases upon contact with water
WGK
WGK 1
Flash Point(F)
64.4 °F
Flash Point(C)
18 °C
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Highly catalytic enantioselective reduction of aromatic ketones using chiral polymer-supported Corey, Bakshi, and Shibata catalysts
Degni S, et al.
Tetrahedron Asymmetry, 15, 1495-1499 (2004)
Efficient synthesis of 1, 3, 5-oxygenated synthons from dimethyl 3-oxoglutarate: first use of borane-dimethyl sulfide complex as a regioselective reducing agent of 3-oxygenated glutarate derivatives
Riatto VB, et al.
Journal of the Brazilian Chemical Society, 22(1), 172-175 (2011)
Study of Hydroboration of (μ-H)2Os3(CO)10 with Various Borane Complexes(BH3?L: L=Lewis base)
Chung JH
J. Korean Chem. Soc., 47, 675-678 (2003)
Generation of bis (pentafluorophenyl) borane-dimethyl sulfide complex as a solution of hexane and its application to hydroboration of alk-1-yne with pinacolborane
Hoshi M, et al.
Tetrahedron Letters, 48(48), 8475-8478 (2007)
A convenient method for the reduction of ozonides to alcohols with borane-dimethyl sulfide complex
Flippin LA, et al.
The Journal of Organic Chemistry, 54(6), 1430-1432 (1989)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
![9-Borabicyclo[3.3.1]nonane solution 0.5 M in THF](/deepweb/assets/sigmaaldrich/product/structures/180/891/8b64e597-269d-4780-98b6-40889dfd06b9/640/8b64e597-269d-4780-98b6-40889dfd06b9.png)