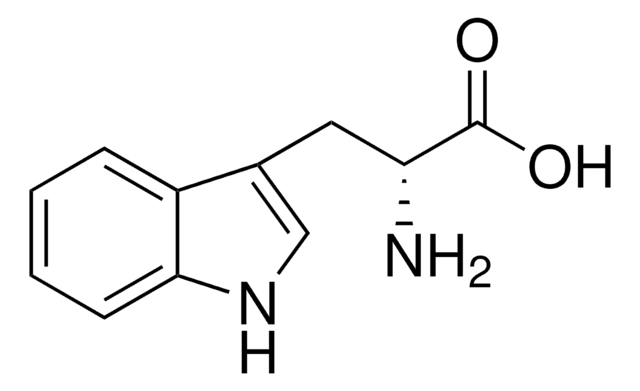
M0534
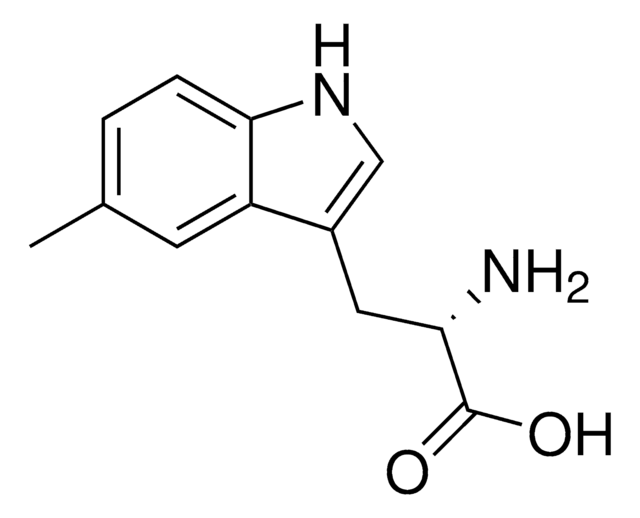
5-Methyl-DL-tryptophan
tryptophan analog
Synonym(s):
5-Methyltryptophan
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Empirical Formula (Hill Notation):
C12H14N2O2
CAS Number:
Molecular Weight:
218.25
Beilstein:
20225
EC Number:
MDL number:
UNSPSC Code:
12352209
PubChem Substance ID:
NACRES:
NA.26
Recommended Products
Quality Level
Assay
≥97% (TLC)
form
powder
color
white to faint yellow
mp
280-282 °C
application(s)
peptide synthesis
storage temp.
2-8°C
SMILES string
Cc1ccc2[nH]cc(CC(N)C(O)=O)c2c1
InChI
1S/C12H14N2O2/c1-7-2-3-11-9(4-7)8(6-14-11)5-10(13)12(15)16/h2-4,6,10,14H,5,13H2,1H3,(H,15,16)
InChI key
HUNCSWANZMJLPM-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- Amelioration of colitis through modulation of gut microbiota: The metabolite 5-Methyl-ᴅʟ-tryptophan (5-MT), derived from Angelica sinensis polysaccharides, was found to ameliorate colitis by modulating gut microbiota and the TLR4/MyD88/NF-κB signaling pathway. This suggests a potential application of 5-MT in inflammatory bowel disease research and therapy (Zou et al., 2023).
- Enzymatic synthesis of tryptophan derivatives: A study on the one-pot enantioselective synthesis of (S)-spirobrassinin and non-natural (S)-methylspirobrassinin from amino acids highlights a method using a turnip enzyme. This research outlines a novel approach to synthesizing tryptophan derivatives, potentially useful in biochemical assays (Ryu et al., 2021).
Biochem/physiol Actions
5-Methyl-DL-tryptophan inhibits the synthesis of anthranilate compounds that are the first steps in the biosynthesis of tryptophan in Neurospora crassa. 5-Methyl-DL-tryptophan is a corepressor of the E. coli trp repressor.
5-Methyl-DL-tryptophan may be used to select genetic mutants of PS strain of Methanococcus voltae (archaebacteria). 5-Methyl-tryptophan is a repressor trp operon expression. 5-Methyl-tryptophan is a substrate for tryptophanase. 5-Methyl-tryptophan inhibits the induction of anthranilate synthase activity by elicitors in oats.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
X H Zhang et al.
Plant physiology, 127(1), 131-141 (2001-09-13)
Anthranilate synthase (AS), the control enzyme of the tryptophan (Trp) biosynthetic pathway, is encoded by nuclear genes, but is transported into the plastids. A tobacco (Nicotiana tabacum) cDNA (ASA2) encoding a feedback-insensitive tobacco AS alpha-subunit was transformed into two different
G Lester
Journal of bacteriology, 96(5), 1768-1773 (1968-11-01)
The in vivo regulation of intermediate reactions in the pathway of tryptophan synthesis in Neurospora crassa was examined in a double mutant (tr-2, tr-3) which lacks the functions of the first and last enzymes in the pathway from chorismic acid
Vered Tzin et al.
The New phytologist, 194(2), 430-439 (2012-02-03)
The shikimate pathway of plants mediates the conversion of primary carbon metabolites via chorismate into the three aromatic amino acids and to numerous secondary metabolites derived from them. However, the regulation of the shikimate pathway is still far from being
Tetsuya Matsukawa et al.
Zeitschrift fur Naturforschung. C, Journal of biosciences, 57(1-2), 121-128 (2002-04-03)
Oat phytoalexins, avenanthramides, are a series of substituted hydroxycinnamic acid amides with anthranilate. The anthranilate in avenanthramides is biosynthesized by anthranilate synthase (AS, EC 4.1.3.27). Induction of anthranilate synthase activity was investigated in oat leaves treated with oligo-N-acetylchitooligosaccharide elicitors. AS
E I Hyde et al.
European journal of biochemistry, 201(3), 569-579 (1991-11-01)
The Escherichia coli trp repressor binds to the trp operator in the presence of tryptophan, thereby inhibiting tryptophan biosynthesis. Tryptophan analogues lacking the alpha-amino group act as inducers of trp operon expression. We have used one- and two-dimensional 1H-NMR spectroscopy
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service