H9903
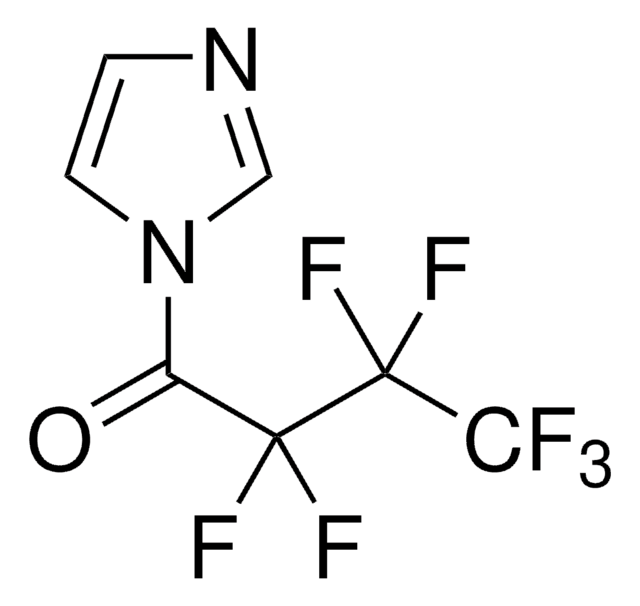
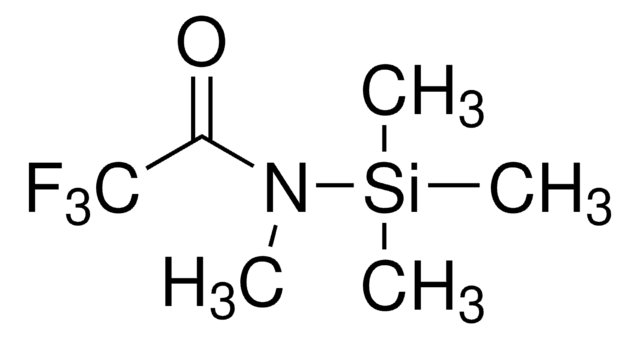
1-(Heptafluorobutyryl)imidazole
BioReagent, suitable for derivatization
Synonym(s):
1-(Perfluorobutyryl)imidazole
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C7H3F7N2O
CAS Number:
Molecular Weight:
264.10
Beilstein:
4488026
EC Number:
MDL number:
UNSPSC Code:
12352005
PubChem Substance ID:
NACRES:
NA.32
Recommended Products
product line
BioReagent
Quality Level
bp
158-163 °C (lit.)
suitability
suitable for derivatization
storage temp.
−20°C
SMILES string
FC(F)(F)C(F)(F)C(F)(F)C(=O)n1ccnc1
InChI
1S/C7H3F7N2O/c8-5(9,6(10,11)7(12,13)14)4(17)16-2-1-15-3-16/h1-3H
InChI key
MSYHGYDAVLDKCE-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
1-(Heptafluorobutyryl)imidazole, also known as HFBI, is an acylating derivatization reagent. It is a mild amine-group derivatizing agent. HFBI doesn’t produce acidic by-products in reaction, and the unutilized reagent doesn’t damage the chromatographic process. Post derivatization reaction, the HFB (Heptafluorobutyryl) derivatives are analyzed chromatographically.
Application
1-(Heptafluorobutyryl)imidazole is used as a derivatizing agent for analysing Sulphur mustard metabolites. It is suitable to study dispersive derivatization of degradation products.
Biochem/physiol Actions
Mild amine-group derivatizing reagent; non-acidic by-product prevents decomposition and reduces GC column degradation.
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
170.6 °F - closed cup
Flash Point(C)
77 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
The use of tandem mass spectrometry for the identification and quantitation of tryptolines (tetrahydro-beta-carbolines) in tissue extracts.
J V Johnson et al.
Progress in clinical and biological research, 183, 161-177 (1985-01-01)
Zhiyong Nie et al.
Talanta, 85(2), 1154-1159 (2011-07-06)
The N-terminal valine adduct (HETE-Val) in globin is believed to behave as a long-lived biomarker after exposure to sulfur mustard (HD). Development of a highly sensitive method for monitoring HETE-Val, particularly at low HD exposure levels or for retrospective detection
Minjia Huang et al.
Analytical sciences : the international journal of the Japan Society for Analytical Chemistry, 21(11), 1343-1347 (2005-12-02)
Headspace solid-phase microextraction (HS-SPME) coupled with gas chromatography/mass spectrometry (GC/MS) method was developed to determine 3-chloropropane-1,2-diol (3-MCPD) in hydrolyzed vegetable protein and Chinese soy sauce. The 3-MCPD was firstly derivativized with phenylboronic acid in aqueous solution at 90 degrees C
Jussi J Joensuu et al.
Methods in molecular biology (Clifton, N.J.), 824, 527-534 (2011-12-14)
Two main hurdles hinder the widespread acceptance of plants as a preferred protein expression platform: low accumulation levels and expensive chromatographic purification methods. Fusion of proteins of interest to fungal hydrophobins has provided a tool to address both accumulation and
A Fidder et al.
Archives of toxicology, 74(4-5), 207-214 (2000-08-26)
The development of a procedure for retrospective detection and quantitation of exposure to the arsenical dichloro(2-chlorovinyl)arsine (lewisite; L1) has been initiated. Upon incubation of human blood with [14C]L1 (20 nM-0.2 mM) in vitro, more than 90% of the total radioactivity
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service