D6566
Dihydrofolate Reductase human
≥80% (SDS-PAGE), recombinant, expressed in E. coli, ≥1 units/mg protein
Synonym(s):
DHFR, Tetrahydrofolate NADP+ oxidoreductase
About This Item
Recommended Products
recombinant
expressed in E. coli
Quality Level
Assay
≥80% (SDS-PAGE)
form
solution
specific activity
≥1 units/mg protein
mol wt
25 kDa
concentration
0.02-0.06 mg/mL
UniProt accession no.
shipped in
wet ice
storage temp.
−20°C
Gene Information
human ... DHFR(1719)
General description
Application
- to investigate the stable expression of green fluorescent protein and the targeted disruption of thioredoxin peroxidase-1 gene in Babesia bovis
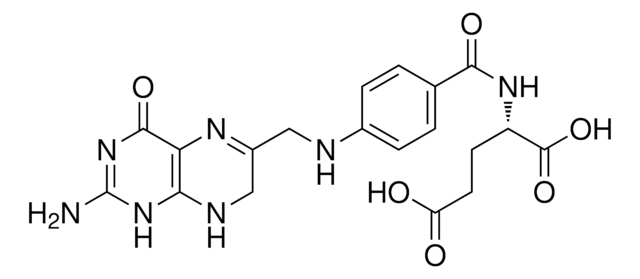
- to study the structural analysis of human dihydrofolate reductase as a binary complex
- to study its in vitro kinetic assay for the enzyme inhibition study
Biochem/physiol Actions
NADPH 0.16 mM
7,8-dihydrofolate 0.03 mM
8-methylpterin 0.13 mM
Ki7
Folate 2.6x10-5 mM
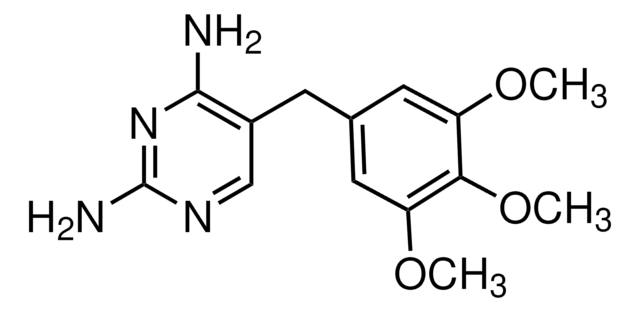
Methotrexate 6.1-9x10-9
Unit Definition
Physical form
substrate
Storage Class Code
12 - Non Combustible Liquids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
This article reviews some of our newest and most innovative technologies and their specific applications toward cancer research. It describes how complex the disease of cancer is, and how difficult it is to identify one topic that is completely unrelated to any other.
This issue of Biofiles reviews some of our newest and most innovative technologies and their specific applications toward cancer research. In preparing this issue of Biofiles, one is reminded how complex the disease of cancer is, and how difficult it is to identify one topic that is completely unrelated to any other.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service