37879
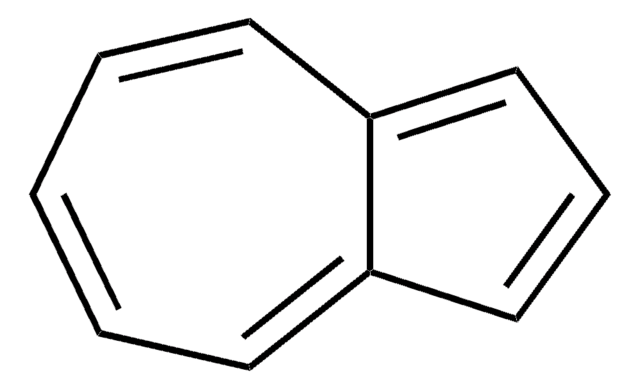
Azulene
analytical standard
Synonym(s):
Bicyclo[5.3.0]decapentaene
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C10H8
CAS Number:
Molecular Weight:
128.17
Beilstein:
969517
EC Number:
MDL number:
UNSPSC Code:
41116107
PubChem Substance ID:
NACRES:
NA.24
Recommended Products
grade
analytical standard
shelf life
limited shelf life, expiry date on the label
technique(s)
HPLC: suitable
gas chromatography (GC): suitable
bp
242 °C (lit.)
mp
98-100 °C (lit.)
application(s)
environmental
format
neat
SMILES string
c1ccc2cccc2cc1
InChI
1S/C10H8/c1-2-5-9-7-4-8-10(9)6-3-1/h1-8H
InChI key
CUFNKYGDVFVPHO-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Refer to the product′s Certificate of Analysis for more information on a suitable instrument technique. Contact Technical Service for further support.
Recommended products
Find a digital Reference Material for this product available on our online platform ChemisTwin® for NMR. You can use this digital equivalent on ChemisTwin® for your sample identity confirmation and compound quantification (with digital external standard). An NMR spectrum of this substance can be viewed and an online comparison against your sample can be performed with a few mouseclicks. Learn more here and start your free trial.
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Chronic 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Lorenzo Stella et al.
Chemical communications (Cambridge, England), (39)(39), 4744-4746 (2008-10-03)
We show here that the recently reported surprisingly large association constant (K = 7.6 x 10(4) M(-1)) between azulene and [60]fullerene is due to experimental artifacts, pointing out potential errors in the characterization of association equilibria by fluorescence spectroscopy, and
Aguinaldo Silva Garcez et al.
Oral surgery, oral medicine, oral pathology, oral radiology, and endodontics, 102(4), e93-e98 (2006-09-26)
To investigate the action of a red laser associated with a photosensitizer on the reduction of Enterococcus faecalis in dental root canal in vitro. Thirty prepared teeth with single canals were contaminated. The chemical group was irrigated with 0.5% NaOCl
Stefan Löber et al.
Bioorganic & medicinal chemistry letters, 22(23), 7151-7154 (2012-10-27)
Based on the dopamine D(4) receptor partial agonist FAUC 3019, a series of azulenylmethylpiperazines was synthesized and affinities for the monoaminergic GPCRs including dopamine, serotonin, histamine and α-adrenergic receptor subtypes were determined. Ligand efficacies of the most promising test compounds
Mine Ince et al.
Chemical communications (Cambridge, England), 48(34), 4058-4060 (2012-03-22)
A novel supramolecular electron donor-acceptor hybrid (2·1) and an electron donor-acceptor conjugate (3), both exhibiting a remarkably shifted Q band in the NIR region of the solar spectrum, were prepared. Irradiation of the supramolecular ensemble 2·1 within the visible range
Dawei Zhao et al.
The Journal of chemical physics, 131(18), 184307-184307 (2009-11-18)
The infrared (IR) spectrum of protonated azulene (AzuH(+), C(10)H(9)(+)) has been measured in the fingerprint range (600-1800 cm(-1)) by means of IR multiple photon dissociation (IRMPD) spectroscopy in a Fourier transform ion cyclotron resonance mass spectrometer equipped with an electrospray
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service