All Photos(1)
About This Item
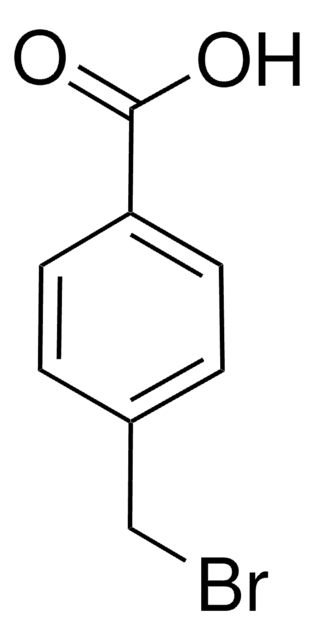
Linear Formula:
C6H5CH2OC6H4CO2H
CAS Number:
Molecular Weight:
228.24
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
99%
mp
189-192 °C (lit.)
functional group
carboxylic acid
phenyl
SMILES string
OC(=O)c1ccc(OCc2ccccc2)cc1
InChI
1S/C14H12O3/c15-14(16)12-6-8-13(9-7-12)17-10-11-4-2-1-3-5-11/h1-9H,10H2,(H,15,16)
InChI key
AQSCHALQLXXKKC-UHFFFAOYSA-N
General description
4-Benzyloxybenzoic acid is a substituted benzoic acid that can be prepared by the benzylation of 4-hydroxybenzoic acid with benzyl bromide.
Application
4-Benzyloxybenzoic acid may be used in the preparation of 1,3-phenylene bis(4-benzyloxybenzoate). It may also be used in the preparation of (-)-(2R,3R)-5,7-bis(benzyloxy)-2-[3,4,5-tris(benzyloxy)-phenyl]chroman-3-yl-(4-benzyloxy)benzoate.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Synthesis and characterization of achiral banana-shaped liquid crystalline molecules containing bisnaphthyl moieties.
Yang PJ and Lin HC.
Liq. Cryst., 33(5), 587-603 (2006)
Henryk Marona et al.
Acta poloniae pharmaceutica, 60(6), 477-480 (2004-04-15)
A series of alkanolamides have been tested for anticonvulsant activity in the maximal electroshock seizure (MES) and subcutaneous pentylenetetrazole seizure treshold (ScMet) assays and for neurotoxicity (TOX) in rodents. Most interesting were the anticonvulsant results of 2N-methylaminoethanol derivative II, which
Kumi Osanai et al.
Tetrahedron, 63(32), 7565-7570 (2007-08-06)
The total and semi syntheses of (2R, 3R)-epigallocatechin-3-O-(4-hydroxybenzoate), a novel catechin from Cistus salvifolius, was accomplished. The proteasome inhibition and cytotoxic activities of the synthetic compound and its acetyl derivative were studied and compared with (2R, 3R)-epigallocatechin-3-gallate (EGCG), the active
Semi-synthesis and proteasome inhibition of D-ring deoxy analogs of (-)-epigallocatechin gallate (EGCG), the active ingredient of green tea extract.
Huo C, et al.
Canadian Journal of Chemistry, 86(6), 495-502 (2008)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service