222380
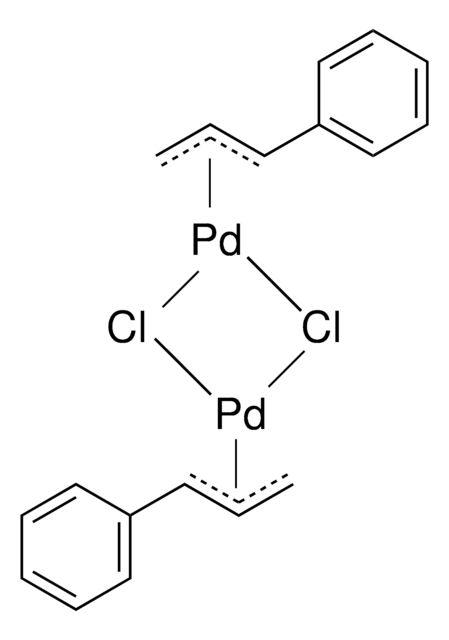
Allylpalladium(II) chloride dimer
98%
Synonym(s):
Bis((η3-allyl)(chloro)palladium), Di-ο-allyldi-μ-chlorodipalladium, [PdCl(C3H5)]2
About This Item
Recommended Products
Quality Level
Assay
98%
form
solid
reaction suitability
core: palladium
reaction type: Cross Couplings
reagent type: catalyst
reaction type: C-H Activation
greener alternative product characteristics
Catalysis
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
greener alternative category
, Aligned
storage temp.
2-8°C
SMILES string
Cl[Pd]CC=C.Cl[Pd]CC=C
InChI
1S/2C3H5.2ClH.2Pd/c2*1-3-2;;;;/h2*3H,1-2H2;2*1H;;/q;;;;2*+1/p-2
InChI key
TWKVUTXHANJYGH-UHFFFAOYSA-L
Looking for similar products? Visit Product Comparison Guide
General description
Application
- Synthesis of cationic palladium cataysts, used in the microwave-assisted Heck arylation.
- Synthesis of N-heterocyclic carbene-palladium-η3-allyl chloride complexes, which are efficient catalyst for the Suzuki-Miyaura cross-coupling of aryl bromides and activated aryl chlorides.
- Synthesis of 1,4-diallyl-1,2-dihydroisoquinolines.
- As catalyst for greener Buchwald-Hartwig coupling in TPGS-750-M.
On the Way Towards Greener Transition-Metal-Catalyzed Processes as Quantified by E Factors
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
TPGS-750-M, a second generation surfactant, is useful for room temperature, palladium and ruthenium-catalyzed reactions in water. Reactions include the Heck reaction, Suzuki-Miyaura reaction, Sonogashira reaction, Buchwald-Hartwig amination reaction, Negishi reaction, and olefin metathesis.
The Heck reaction is the palladium catalyzed cross-coupling reaction between alkenes and aryl or vinyl halides (or triflates) to afford substituted alkenes.
Protocols
Buchwald-Hartwig Amination Reaction in Water at Room Temperature using TPGS-750-M
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service![[Pd(allyl)Cl]2 Umicore](/deepweb/assets/sigmaaldrich/product/structures/367/851/7e957f32-7c31-40bf-8349-77de7cc990e4/640/7e957f32-7c31-40bf-8349-77de7cc990e4.png)



![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)

![Di-μ-mesylbis[2′-(amino-N)[1,1′-biphenyl]-2-yl-C]dipalladium(II) 95%](/deepweb/assets/sigmaaldrich/product/structures/197/956/09a1e75d-7d4a-407a-91e9-982e811baf0b/640/09a1e75d-7d4a-407a-91e9-982e811baf0b.png)