1176007
USP
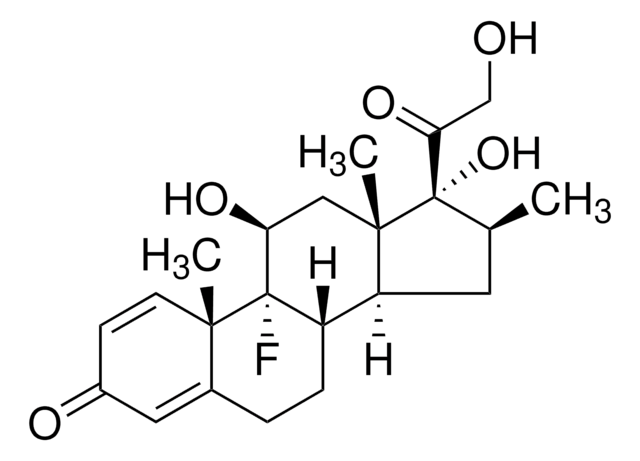
Dexamethasone
United States Pharmacopeia (USP) Reference Standard
Synonym(s):
(11β,16α)-9-Fluoro-11,17,21-trihydroxy-16-methylpregna-1,4-diene-3,20-dione, 9α-Fluoro-16α-methyl-11β,17α,21-trihydroxy-1,4-pregnadiene-3,20-dione, 9α-Fluoro-16α-methylprednisolone, Prednisolone F
About This Item
Recommended Products
biological source
synthetic
grade
pharmaceutical primary standard
Agency
USP
vapor pressure
<0.0000001 kPa ( 25 °C)
API family
dexamethasone
packaging
pkg of 125 mg
manufacturer/tradename
USP
color
white
mp
262-264 °C (lit.)
solubility
acetone: sparingly soluble
chloroform: slightly soluble
ethanol: sparingly soluble
ether: very slightly soluble
methanol: sparingly soluble
water: practically insoluble
application(s)
pharmaceutical (small molecule)
format
neat
storage temp.
2-8°C
SMILES string
C[C@@H]1C[C@H]2[C@@H]3CCC4=CC(=O)C=C[C@]4(C)[C@@]3(F)[C@@H](O)C[C@]2(C)[C@@]1(O)C(=O)CO
InChI
1S/C22H29FO5/c1-12-8-16-15-5-4-13-9-14(25)6-7-19(13,2)21(15,23)17(26)10-20(16,3)22(12,28)18(27)11-24/h6-7,9,12,15-17,24,26,28H,4-5,8,10-11H2,1-3H3/t12-,15+,16+,17+,19+,20+,21+,22+/m1/s1
InChI key
UREBDLICKHMUKA-CXSFZGCWSA-N
Gene Information
human ... NR3C1(2908)
Looking for similar products? Visit Product Comparison Guide
General description
Application
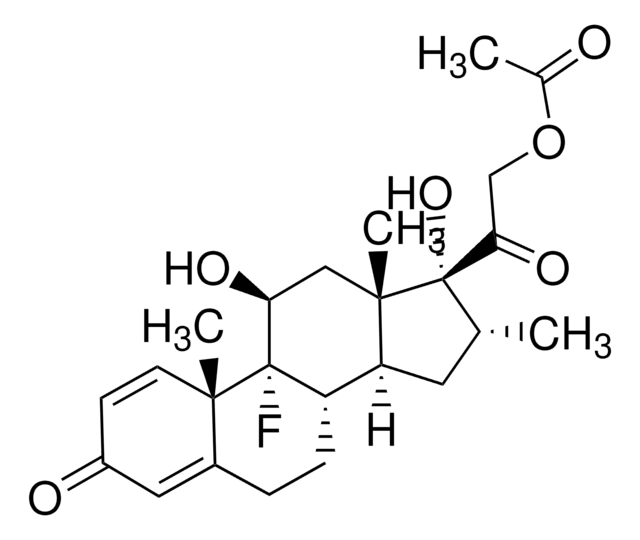
- Ciprofloxacin and Dexamethasone Otic Suspension
- Dexamethasone
- Dexamethasone Compounded Oral Suspension
- Dexamethasone Elixir
- Dexamethasone Injection
- Dexamethasone Ophthalmic Suspension
- Dexamethasone Oral Solution
Biochem/physiol Actions
Analysis Note
Other Notes
related product
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Repr. 1B
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Protocols
A simple, precise and sensitive Reverse-Phase High Pressure Liquid Chromatography gradient method was adapted for traceability, homogeneity and total chromatographic analysis of Dexamethasone. The given experimental conditions follow the USP43-NF38 monograph method for Dexamethasone Assay and Organic Impurity Profiling. Dexamethasone, Betamethasone, Dexamethasone acetate and Desoximetasone were baseline resolved within 20 minutes using a Titan C18 UHPLC column (2.1 x 100 mm, 1.9 µm).
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service