E0162
Carboxylesterase 1 isoform c human
recombinant, expressed in baculovirus infected BTI insect cells
Synonym(s):
Carboxylesterase 1 human, carboxylesterase, esterase
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Recommended Products
recombinant
expressed in baculovirus infected BTI insect cells
Quality Level
form
liquid
concentration
≥0.3 mg/mL
shipped in
dry ice
storage temp.
−70°C
Looking for similar products? Visit Product Comparison Guide
General description
Carboxylesterase 1 (CE1) is a member of a large multigene carboxylesterase α,β-hydrolase family. It is majorly expressed in the liver. CE1 comprises an αβ domain, a central catalytic domain and a regulatory domain.
Application
Carboxylesterase 1 isoform c human has been used as a reference standard in carboxylesterase activity from the mussel for comparison of substrate specificity and inhibition studies. It has also been used as a commercial recombinant protein for the methodological validation of environmental chemical-based inhibition studies.
Biochem/physiol Actions
Carboxylesterase enzymes are responsible for the hydrolysis of ester- and amide-bond-containing drugs such as cocaine and heroin. They also hydrolyze long-chain fatty acid esters and thioesters. Carboxylesterase 1 (CE1) catalyzes the formation of cholesteryl esters from cholesterol and fatty acids. Through a transesterification reaction, CE1 also mediates the generation of fatty acid ethyl esters (FAEEs). It also hydrolyzes aromatic and aliphatic esters with preference to small alcohol groups and bulky acyl groups. CE1 metabolizes drug esters and amides carbamates. It participates in the detoxification of environmental toxicants and carcinogens and is useful in pharmacokinetic studies for evaluating pro-drugs.
Physical properties
This product is offered in a volume of 0.5 mL.
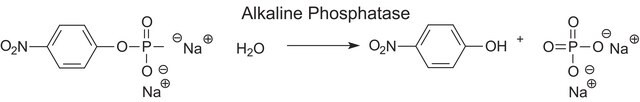
Unit Definition
One unit will hydrolyze one nanomole of 4-nitrophenyl acetate per minute at pH 7.4 at 37 °C.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Resp. Sens. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Montserrat Solé et al.
Environmental toxicology and pharmacology, 82, 103561-103561 (2020-12-12)
Carboxylesterases (CEs) are key enzymes which catalyse the hydrolysis reactions of multiple xenobiotics and endogenous ester moieties. Given their growing interest in the context of marine pollution and biomonitoring, this study focused on the in vitro sensitivity of marine invertebrate
Jihong Lian et al.
Protein & cell, 9(2), 178-195 (2017-07-06)
Mammalian carboxylesterases hydrolyze a wide range of xenobiotic and endogenous compounds, including lipid esters. Physiological functions of carboxylesterases in lipid metabolism and energy homeostasis in vivo have been demonstrated by genetic manipulations and chemical inhibition in mice, and in vitro
Masakiyo Hosokawa
Molecules (Basel, Switzerland), 13(2), 412-431 (2008-02-29)
Mammalian carboxylesterases (CESs) comprise a multigene family whose gene products play important roles in biotransformation of ester- or amide-type prodrugs. They are members of an alpha,beta-hydrolase-fold family and are found in various mammals. It has been suggested that CESs can
Dandan Wang et al.
Acta pharmaceutica Sinica. B, 8(5), 699-712 (2018-09-25)
Mammalian carboxylesterases (CEs) are key enzymes from the serine hydrolase superfamily. In the human body, two predominant carboxylesterases (CES1 and CES2) have been identified and extensively studied over the past decade. These two enzymes play crucial roles in the metabolism
B Sànchez-Nogué et al.
Environmental science and pollution research international, 20(5), 3480-3488 (2012-12-06)
The common sole, Solea solea (Linneus, 1758), and the Senegalese sole, Solea senegalensis (Kaup, 1858), are two important commercial species that coexist in the NW Mediterranean. In order to assess the species' ability to respond to chemical insults, a comparison
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service