750271
Potassium trans-1-propenyltrifluoroborate
97%
Synonym(s):
Potassium (E)-trifluoro(1-propenyl)borate
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
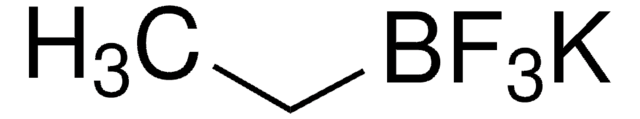
Empirical Formula (Hill Notation):
C3H5BF3K
CAS Number:
Molecular Weight:
147.98
MDL number:
UNSPSC Code:
12352103
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
form
solid
mp
81-85 °C
SMILES string
[K+].C\C=C\[B-](F)(F)F
InChI
1S/C3H5BF3.K/c1-2-3-4(5,6)7;/h2-3H,1H3;/q-1;+1/b3-2+;
InChI key
RLDWVFWDURMTAV-SQQVDAMQSA-N
Application
Potassium trans-1-propenyltrifluoroborate can be used:
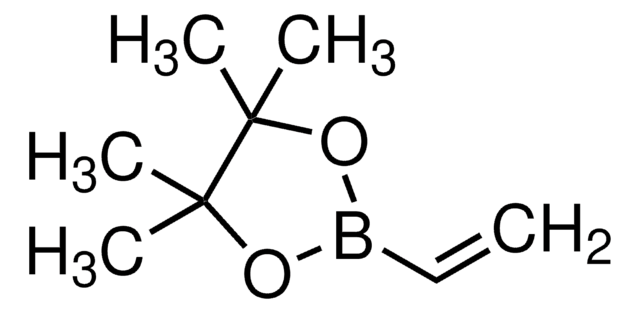
- As a substrate in the cross-coupling reactions with 2-(chloromethyl)-2,1-borazaronaphthalenes to yield allyl borazaronaphthalenes using a palladium catalyst.
- In the synthesis of anethole dithiolethione-NH2 (ADT-NH2), which in turn is used to prepare a drug-H2S delivery system.
- To prepare methyl (E)-2,2-dimethyl-5-(propen-1-yl)-4H-benzo[d][1,3]dioxine-7-carboxylate, a key intermediate for the synthesis of the alkaloid ampullosine.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Accessing 2-(Hetero) arylmethyl-,-allyl-, and-propargyl-2, 1-borazaronaphthalenes: Palladium-Catalyzed Cross-Couplings of 2-(Chloromethyl)-2, 1-borazaronaphthalenes
Molander GA, et al.
Organic Letters, 16(22), 6024-6027 (2014)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service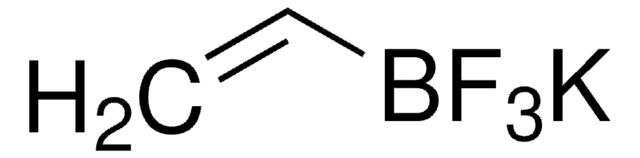
![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)
![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II), complex with dichloromethane](/deepweb/assets/sigmaaldrich/product/structures/825/986/4317978b-1256-4c82-ab74-6a6a3ef948b1/640/4317978b-1256-4c82-ab74-6a6a3ef948b1.png)