274240
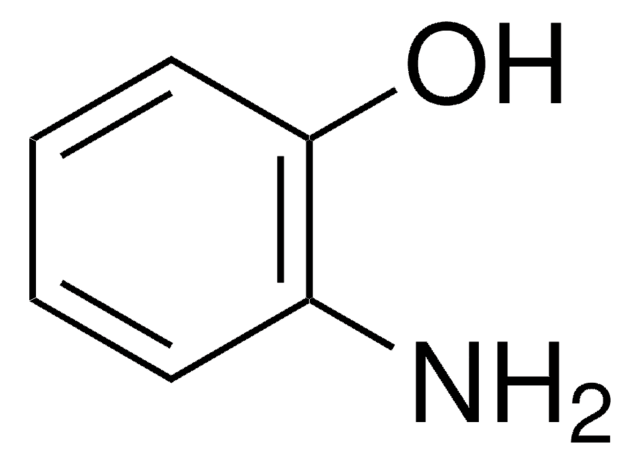
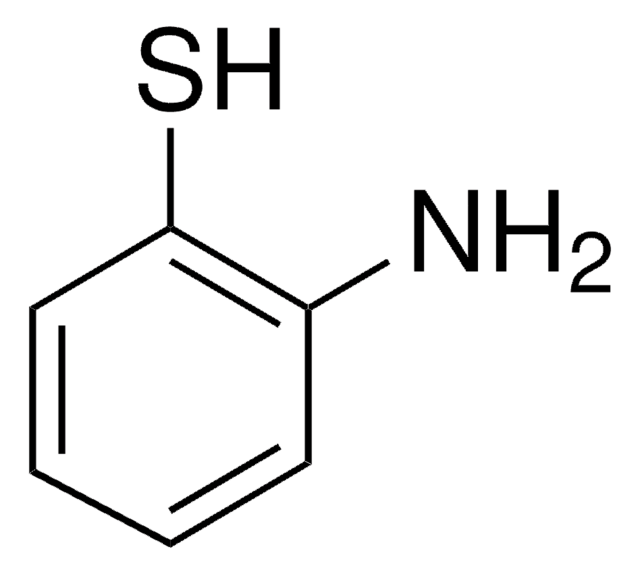
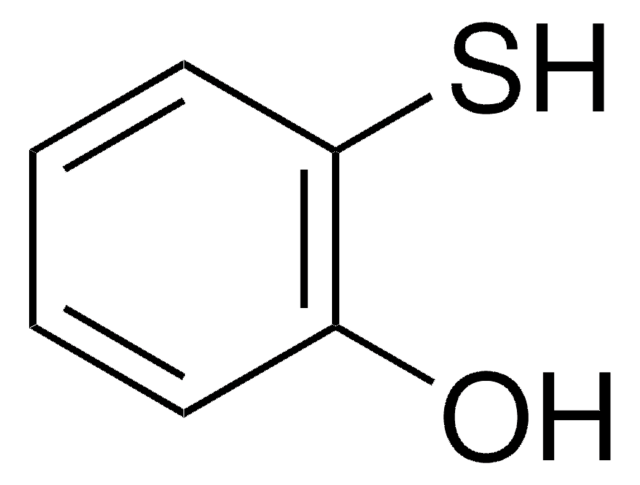
2-Aminothiophenol
99%
Synonym(s):
2-Aminobenzenethiol, 2-Aminophenyl mercaptan, 2-Mercaptoaniline
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
H2NC6H4SH
CAS Number:
Molecular Weight:
125.19
Beilstein:
606076
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
99%
form
liquid
refractive index
n20/D 1.642 (lit.)
bp
70-72 °C/0.2 mmHg (lit.)
mp
16-20 °C (lit.)
density
1.17 g/mL at 25 °C (lit.)
SMILES string
Nc1ccccc1S
InChI
1S/C6H7NS/c7-5-3-1-2-4-6(5)8/h1-4,8H,7H2
InChI key
VRVRGVPWCUEOGV-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
2-Aminothiophenol is an organosulfur compound used in the synthesis of urea derivatives.
Application
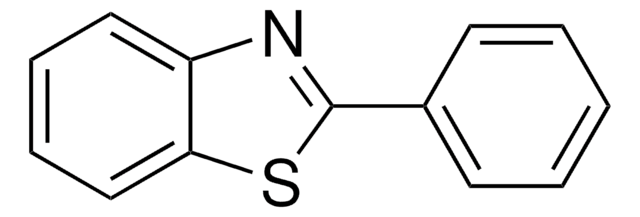
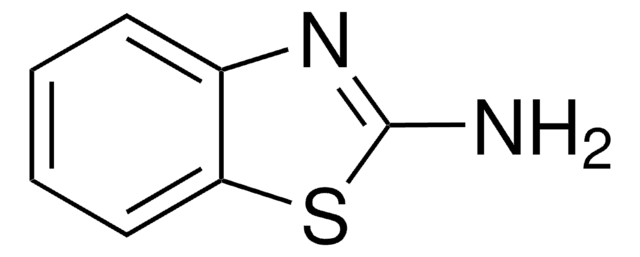
2-Aminothiophenol was used in the synthesis of 1,5-benzothiazepines derivatives by reacting with 1,3-diphenylpropenone derivatives and using aluminosilicate solid catalysts. It was also used in the synthesis of benzothiazole and 3-(benzothiazol-2-yl)coumarin derivatives.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Aquatic Acute 1 - Eye Dam. 1 - Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
174.2 °F
Flash Point(C)
79 °C
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Tianzhi Yu et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 108, 274-279 (2013-03-19)
Two different types of fluorescent materials containing benzothiazolyl moiety, 2-(benzothiazol-2-yl)phenol derivatives and 3-(benzothiazol-2-yl)coumarin derivatives, were synthesized synchronously using ethyl cyanoacetate, appropriate aromatic aldehyde and 2-aminothiophenol as the starting materials under the catalysis of benzoic acid by one-pot reaction. This method
Maria J Climent et al.
ChemSusChem, 7(4), 1177-1185 (2014-03-13)
1,5-Benzothiazepines derivatives were obtained first by starting from 1,3-diphenylpropenone derivatives (chalcones) and 2-aminothiophenol by using aluminosilicate solid catalysts. However, diffusional limitations and the strong adsorption of products on the catalyst are deleterious for catalyst activity and life. Then a structured
Vikas S Patil et al.
Journal of fluorescence, 23(5), 1019-1029 (2013-05-18)
Novel ESIPT inspired benzimidazole, benzoxazole and benzothiazole were synthesized from 2,4-dihydroxy benzoic acid and 1,2-phenelenediamine, 2-aminophenol, and 2-aminothiophenol respectively. The synthesized 2-(2',4'-dihydroxyphenyl) benzimidazole, benzoxazole and benzothiazole are fluorescent and the emission characteristic are very sensitive to the micro-environment. They show
William P Morrow et al.
Archives of biochemistry and biophysics, 631, 66-74 (2017-08-23)
Thiol dioxygenases are non-heme mononuclear iron enzymes that catalyze the O
Ling-Jie Kong et al.
Analytical and bioanalytical chemistry, 404(6-7), 1653-1660 (2012-07-24)
A simple electrochemical sensor based on a molecularly imprinted polymer film as the recognition element was developed for ractopamine (RAC) detection. This is the first report of a RAC-imprinted film on a gold electrode surface, synthesized through an electrochemical method
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service