225649
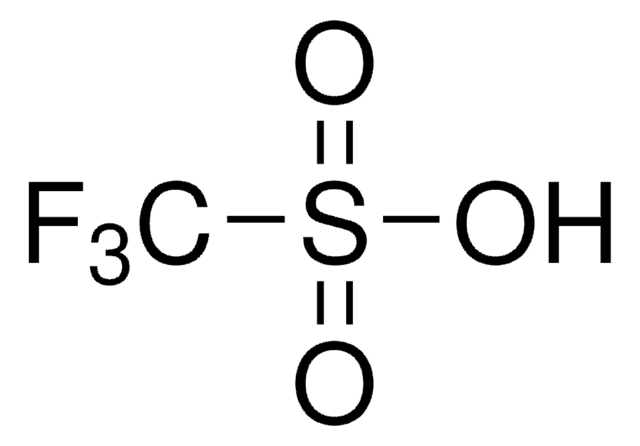
Trimethylsilyl trifluoromethanesulfonate
99%
Synonym(s):
TMS triflate, TMSOTf, Trifluoromethanesulfonic acid trimethylsilylester
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
CF3SO3Si(CH3)3
CAS Number:
Molecular Weight:
222.26
Beilstein:
1868911
EC Number:
MDL number:
UNSPSC Code:
12352103
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
99%
form
liquid
refractive index
n20/D 1.36 (lit.)
bp
77 °C/80 mmHg (lit.)
density
1.228 g/mL at 25 °C (lit.)
SMILES string
C[Si](C)(C)OS(=O)(=O)C(F)(F)F
InChI
1S/C4H9F3O3SSi/c1-12(2,3)10-11(8,9)4(5,6)7/h1-3H3
InChI key
FTVLMFQEYACZNP-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Trimethylsilyl trifluoromethanesulfonate has been used in combination with boron trifluoride etherate for the copper-catalyzed asymmetric allylic alkylation (AAA) of allyl bromides, chlorides, and ethers with organolithium reagents in the presence of a chiral ligand.
It can be used:
It may also be used to catalyze:
It can be used:
- As a silylating agent for the synthesis of trimethylsilyl-enol ethers from esters of α-diazoacetoacetic acid.
- To activate benzyl and allyl ethers for the alkylation of sulfides.
- To facilitate the conversion of Diels-Alder adducts of Danishefsky′s diene to cyclohexenones without the formation of methoxy ketone by-product.
- To prepare difluoroboron triflate etherate, a powerful Lewis acid especially in acetonitrile solvent.
- As a reagent in a Dieckmann-like cyclization of ester-imides and diesters.
It may also be used to catalyze:
- Acylation of alcohols with acid anhydrides.
- Reductive coupling of carbonyl compounds with trialkylsilanes to form symmetrical ethers.
- Glycosidation of 4-demethoxydaunomycinones with 1-O-acyl-L-daunosamine derivatives.
accessory
Product No.
Description
Pricing
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Flam. Liq. 3 - Skin Corr. 1B
Supplementary Hazards
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
77.0 °F - closed cup
Flash Point(C)
25 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
A simple method of preparing trimethylsilyl-and tert-butyldimethylsilyl-enol ethers of ?-diazoacetoacetates and their use in the synthesis of a chiral precursor to thienamycin analogs.
Ueda Y, et al.
Canadian Journal of Chemistry, 62(12), 2936-2940 (1984)
Thomas R Hoye et al.
Organic letters, 8(23), 5191-5194 (2006-11-03)
[Structure: see text] Trialkylsilyl triflates effect cyclization of ester-imides such as 2 to produce adducts such as 4a. Trapping of the in situ generated, nucleophilic ketene acetal (cf. 5a) is a key aspect of the transformation. A range of substrates
BF 3? OEt 2 and TMSOTf: A synergistic combination of Lewis acids.
Myers EL, et al.
Chemical Communications (Cambridge, England), 42, 4434-4436 (2006)
Eddie L Myers et al.
Chemical communications (Cambridge, England), (42), 4434-4436 (2006-10-24)
The combination of BF3.OEt2 and TMSOTf gives BF2OTf.OEt2, which is a more powerful Lewis acid than its components and especially effective in CH3CN solvent; the complex formed has been characterised by 1H, 19F, 11B and 31P (using Et3PO as an
Method for sulfide S-benzylation or S-allylation using trimethylsilyl triflate activated benzyl or allyl ethers.
Vedejs E & Eustache J.
The Journal of Organic Chemistry, 46(16), 3353-3354 (1981)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service