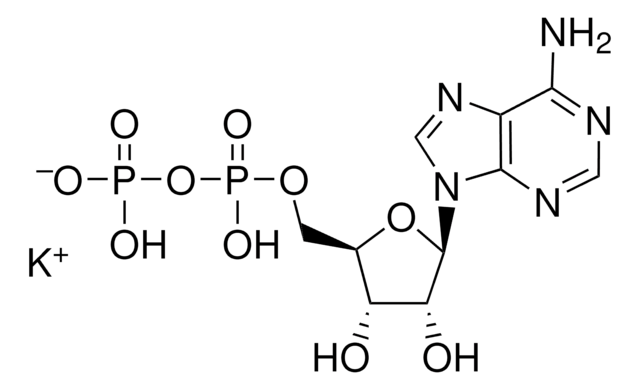
A2810
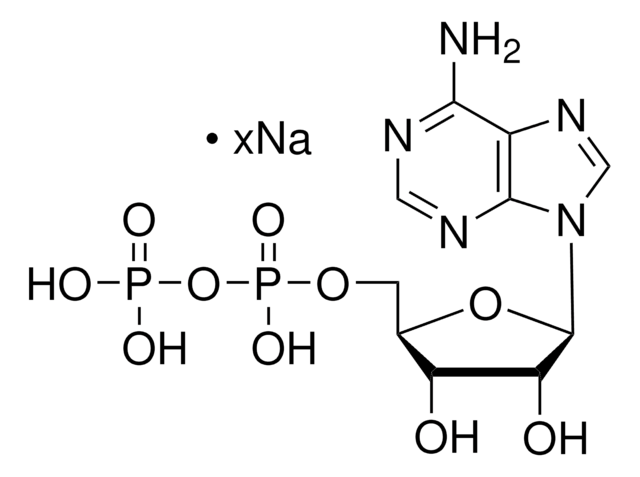
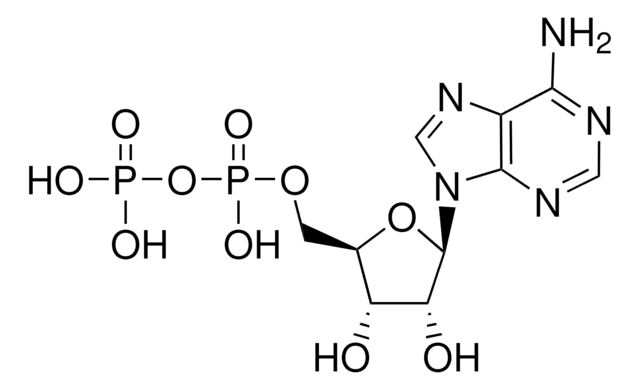
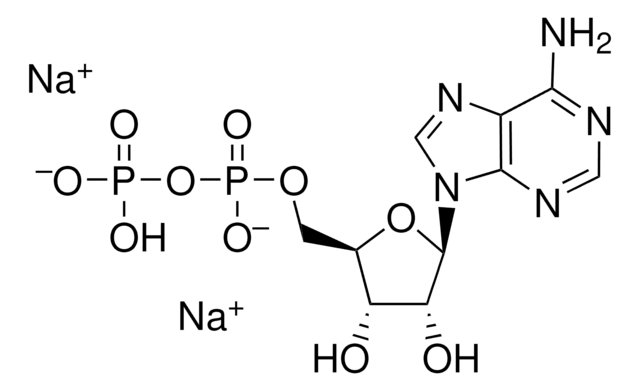
Adenosine 5′-diphosphate–Agarose
lyophilized powder
Synonym(s):
5′-ADP agarose
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Recommended Products
form
lyophilized powder
Quality Level
extent of labeling
1-5 μmol per mL
matrix
cross-linked 4% beaded agarose
matrix activation
cyanogen bromide
matrix attachment
C-8
matrix spacer
9 atoms
storage temp.
−20°C
Looking for similar products? Visit Product Comparison Guide
Application
Adenosine 5′-diphosphate has been used in the research of platelet integrin α(IIb)β3, which is crucial for platelet aggregation. It has been determined that the interaction between Adenosine 5′-diphosphate and the receptor P2Y12 is needed for the maintenance of integrin α(IIb)β3 activation. Adenosine 5′-diphosphate agarose (5′-ADP agarose) has been used in purification of heat schock proteins.
Quantity
Swelling factor = 1 gram yields 8-16 mL packed gel
Physical form
Lyophilized powder stabilized with lactose
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Xueji Wu et al.
The Biochemical journal, 378(Pt 3), 793-799 (2003-12-11)
The chaperone activity of Hsp70 (70 kDa heat-shock protein) in protein folding and its conformational switch, including oligomeric and monomeric interconversion, are regulated by the hydrolysis of ATP and the ATP-ADP exchange cycle. The crystal structure of human ATPase domain
Zihai Li
Methods (San Diego, Calif.), 32(1), 25-28 (2003-11-20)
Known commonly as molecular chaperones for proteins, heat shock proteins (HSPs) have also been found to chaperone small molecular weight cellular peptides. HSP-peptide complexes can prime T cell immunity specific against the peptides bound to HSPs, but not against HSPs
Antoine Ménoret
Methods (San Diego, Calif.), 32(1), 7-12 (2003-11-20)
Heat shock proteins (HSPs) are powerful immunogens against the antigenic peptides they chaperone. The antigenic peptides are MHC I-binding peptides and their elongated precursors derived from tumor antigens, viral antigens, minor histocompatibility antigens, or model antigens. HSP-peptide complexes can immunize
Knut Fälker et al.
Thrombosis and haemostasis, 92(1), 114-123 (2004-06-24)
Stimulating human platelets with thrombin induces the activation of the extracellular signal-regulated kinase 2 (ERK2). We demonstrate that this effect is highly dependent on ADP secretion and P2Y12 receptor signalling. AR-C69931MX (10 microM), a specific antagonist of the Gi-coupled P2Y12
T Kamae et al.
Journal of thrombosis and haemostasis : JTH, 4(6), 1379-1387 (2006-05-19)
Platelet integrin alpha(IIb)beta3 plays a crucial role in platelet aggregation, and the affinity of alpha(IIb)beta3 for fibrinogen is dynamically regulated. Employing modified ligand-binding assays, we analyzed the mechanism by which alpha(IIb)beta3 maintains its high-affinity state. Washed platelets adjusted to 50
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service