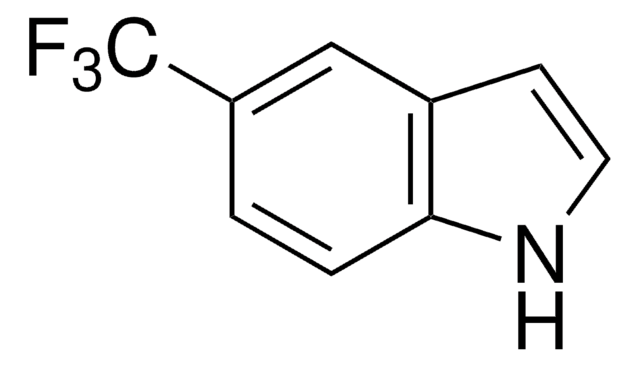
All Photos(1)
About This Item
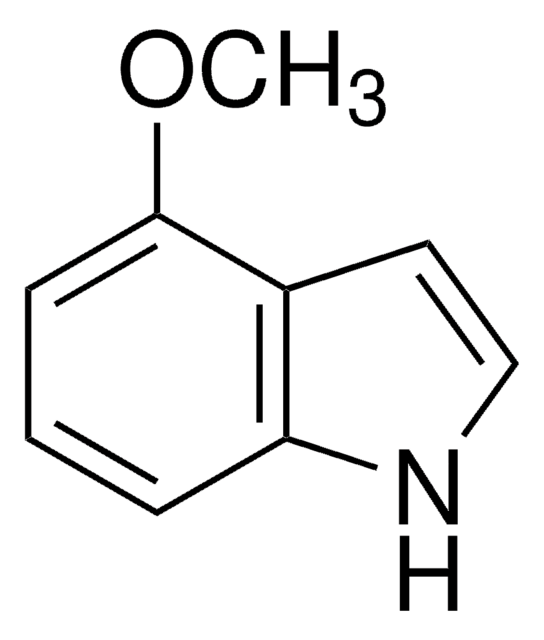
Empirical Formula (Hill Notation):
C9H9NO
CAS Number:
Molecular Weight:
147.17
Beilstein:
116722
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
99%
bp
176-178 °C/17 mmHg (lit.)
mp
52-55 °C (lit.)
SMILES string
COc1ccc2[nH]ccc2c1
InChI
1S/C9H9NO/c1-11-8-2-3-9-7(6-8)4-5-10-9/h2-6,10H,1H3
InChI key
DWAQDRSOVMLGRQ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Christian Brand et al.
The Journal of chemical physics, 133(2), 024303-024303 (2010-07-17)
Rotationally resolved electronic spectra of the vibrationless origin and of eight vibronic bands of 5-methoxyindole (5MOI) have been measured and analyzed using an evolutionary strategy approach. The experimental results are compared to the results of ab initio calculations. All vibronic
Kenichiro Todoroki et al.
Analytical sciences : the international journal of the Japan Society for Analytical Chemistry, 22(2), 281-286 (2006-03-04)
In this paper we describe a highly sensitive and selective liquid chromatographic method for the determination of 5-methoxyindoles (5-methoxyindole-3-acetic acid, 5-methoxytryptamine, 5-methoxytryptophol, and melatonin) using a post-column technique involving electrolytic demethylation followed by fluorescence derivatization with benzylamine. We separated these
Pawel Sledz et al.
Journal of the American Chemical Society, 132(13), 4544-4545 (2010-03-18)
Fragment-based methods are a new and emerging approach for the discovery of protein binders that are potential new therapeutic agents. Several ways of utilizing structural information to guide the inhibitor assembly have been explored to date. One of the approaches
F Peter Guengerich et al.
Journal of medicinal chemistry, 47(12), 3236-3241 (2004-05-28)
Indigoids, a class of bis-indoles, represent a promising protein kinase inhibitor scaffold. Oxidation of indole by cytochrome P450 (P450) has been shown to generate species (indoxyl, isatin) that couple to yield indigo and indirubin. Escherichia coli-expressed human P450 2A6 mutants
Xiang-Qun Hu et al.
The Journal of biological chemistry, 283(11), 6826-6831 (2008-01-12)
Current receptor theory suggests that there is an equilibrium between the inactive (R) and active (R*) conformations of ligand-gated ion channels and G protein-coupled receptors. The actions of ligands in both receptor types could be appropriately explained by this two-state
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service