373753
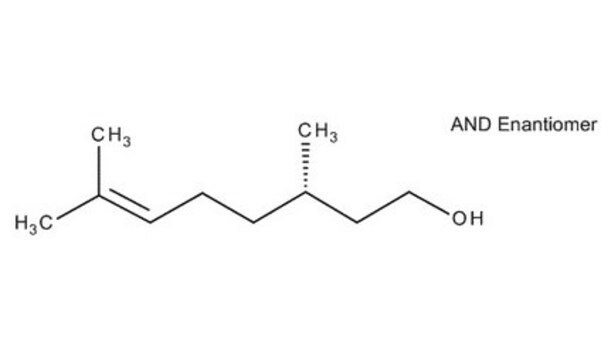
(S)-(−)-Citronellal
96%
Synonym(s):
(−)-Citronellal, (3S)-3,7-Dimethyl-6-octenal
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
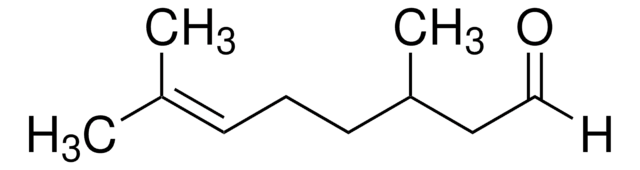
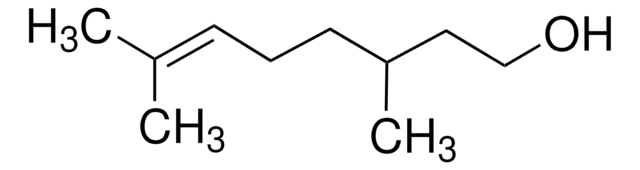
(CH3)2C=CHCH2CH2CH(CH3)CH2CHO
CAS Number:
Molecular Weight:
154.25
Beilstein:
1720790
EC Number:
MDL number:
UNSPSC Code:
12352114
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
96%
form
liquid
optical activity
[α]20/D −15°, neat
refractive index
n20/D 1.446 (lit.)
density
0.851 g/mL at 25 °C (lit.)
functional group
aldehyde
SMILES string
[H]C(=O)C[C@@H](C)CC\C=C(\C)C
InChI
1S/C10H18O/c1-9(2)5-4-6-10(3)7-8-11/h5,8,10H,4,6-7H2,1-3H3/t10-/m0/s1
InChI key
NEHNMFOYXAPHSD-JTQLQIEISA-N
General description
(S)-(-)-Citronellal is a monoterpenoid compound mainly found in Corymbia citriodora and Cymbopogon nardus essential oils.
Application
(S)-(-)-Citronellal may be used in the synthesis of bioactive compounds like (+)-hexahydrocannabinol, (S)-isopulegol, machaeriols A and B.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
167.0 °F - closed cup
Flash Point(C)
75 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Asymmetric cyclization of unsaturated aldehydes catalyzed by a chiral Lewis acid.
Sakane S, et al.
Tetrahedron Letters, 26(45), 5535-5538 (1985)
First synthesis of antimalarial Machaeriols A and B.
Chittiboyina AG, et al.
Tetrahedron Letters, 45(8), 1689-1691 (2004)
Efficient one-pot synthetic approaches for cannabinoid analogues and their application to biologically interesting (-)-hexahydrocannabinol and (+)-hexahydrocannabinol.
Lee YR and Xia L.
Tetrahedron Letters, 49(20), 3283-3287 (2008)
Xiangxian Ying et al.
Molecules (Basel, Switzerland), 24(6) (2019-03-21)
The members of the Old Yellow Enzyme (OYE) family are capable of catalyzing the asymmetric reduction of (E/Z)-citral to (R)-citronellal-a key intermediate in the synthesis of L-menthol. The applications of OYE-mediated biotransformation are usually hampered by its insufficient enantioselectivity and
Osnat Altshuler et al.
Journal of natural products, 76(9), 1598-1604 (2013-08-21)
Citronellal is a major component of Corymbia citriodora and Cymbopogon nardus essential oils. Herein it is shown that whereas (+)-citronellal (1) is an effective microtubule (MT)-disrupting compound, (-)-citronellal (2) is not. Quantitative image analysis of fibroblast cells treated with 1
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service