All Photos(2)
About This Item
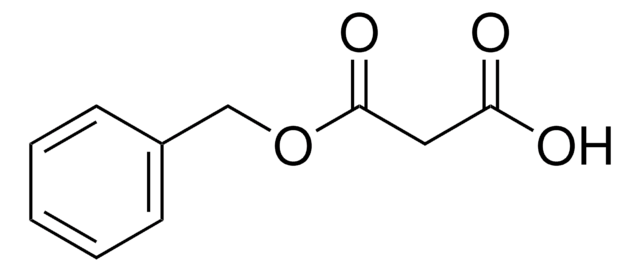
Linear Formula:
CH2(CO2CH2C6H5)2
CAS Number:
Molecular Weight:
284.31
Beilstein:
1998264
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
form
liquid
Quality Level
refractive index
n20/D 1.546 (lit.)
bp
188 °C/0.2 mmHg (lit.)
density
1.137 g/mL at 25 °C (lit.)
functional group
ester
phenyl
SMILES string
O=C(CC(=O)OCc1ccccc1)OCc2ccccc2
InChI
1S/C17H16O4/c18-16(20-12-14-7-3-1-4-8-14)11-17(19)21-13-15-9-5-2-6-10-15/h1-10H,11-13H2
InChI key
RYFCSKVXWRJEOB-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Direct asymmetric reaction of dibenzyl malonate with N-tert-butoxycarbonyl aldimines in the presence of Yb(OTf)3 and iPr-pybox (pybox = pyridine bisoxazoline) complexes has been investigated.
Application
Dibenzyl malonate was used in the preparation of tetraethyl 3,3-bis(benzyloxycarbonyl)propylene bisphosphonate. It was also used in the preparation of benzyl umbelliferone-3-carboxylate via Knoevenagel condensation with 2,4-dihydroxybezaldehyde.
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
230.0 °F - closed cup
Flash Point(C)
110 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Babak Karimi et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 19(31), 10142-10145 (2013-06-22)
Go Mannich! A highly efficient and enantioselective method for the direct asymmetric reaction of dibenzyl malonate with N-tert-butoxycarbonyl aldimines in the presence of Yb(OTf)3 and iPr-pybox complexes is described (see scheme; pybox = pyridine bisoxazoline).
Synthesis of bone-targeted oestrogenic compounds for the inhibition of bone resorption.
Bulman Page PC, et al.
Tetrahedron, 57(9), 1837-1847 (2001)
J D Perry et al.
Journal of applied microbiology, 101(5), 977-985 (2006-10-17)
Enzyme substrates based on 4-methylumbelliferone are widely used for the detection of Escherichia coli and enterococci in water, by detection of beta-glucuronidase and beta-glucosidase activity respectively. This study aimed to synthesize and evaluate novel umbelliferone-based substrates with improved sensitivity for
Prerana D Tomke et al.
Ultrasonics sonochemistry, 38, 496-502 (2017-06-22)
Poly (ethylene glutarate), poly (ethylene malonate) and poly (ethylene phthalate), were enzymatically synthesized by immobilized Candida antarctica lipase B in solvent free conditions. The synthesis of these polyesters was based on the ester-ester exchange reaction between ethylene glycol diacetate and
Riin Rebane et al.
Journal of chromatography. A, 1390, 62-70 (2015-03-12)
Derivatization is one of the most common ways for improving chromatographic separation and sensitivity for LC-ESI-MS analysis. The aim of this work was to design new derivatization reagents for LC-ESI-MS analysis of amino acids which would (1) provide good reversed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
![1,8-Diazabicyclo[5.4.0]undec-7-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)