T9935
Trypsin from bovine pancreas
essentially salt-free, lyophilized powder, ≥9,000 BAEE units/mg protein, BioReagent, suitable for cell culture
Synonym(s):
Serine Protease 1
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Recommended Products
product line
BioReagent
Quality Level
form
essentially salt-free, lyophilized powder
specific activity
≥9,000 BAEE units/mg protein
mol wt
23.8 kDa
composition
protein, ≥80%
technique(s)
cell culture | mammalian: suitable
solubility
hydrochloric acid: soluble 1 mM
storage temp.
−20°C
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
For trypsin digestion of peptides, use a ratio of about 1:100 to 1:20 for trypsin:peptide. The typical use for this product is in removing adherent cells from a culture surface. The concentration of trypsin necessary to dislodge cells from their substrate is dependent primarily on the cell type and the age of the culture. Trypsins have also been used for the re-suspension of cells during cell culture, in proteomics research for digestion of proteins and in various in-gel digestions. Additional applications include assessing crystallization by membrane-based techniques and in a study to determine that protein folding rates and yields can be limited by the presence of kinetic traps.
Trypsin from bovine pancreas has been used for enzymatic digestion of nucleus pulposus (NP) cells during human NP cells isolation. It has also been used as a immunopanning reagent for purification of cells.
Biochem/physiol Actions
Trypsin cleaves peptides on the C-terminal side of lysine and arginine residues. The rate of hydrolysis of this reaction is slowed if an acidic residue is on either side of the cleavage site and hydrolysis is stopped if a proline residue is on the carboxyl side of the cleavage site. The optimal pH for trypsin activity is 7-9. Trypsin can also act to cleave ester and amide linkages of synthetic derivatives of amino acids. EDTA is added to trypsin solutions as a chelating agent that neutralizes calcium and magnesium ions that obscure the peptide bonds on which trypsin acts. Removing these ions increases the enzymatic activity.
Serine protease inhibitors, including DFP, TLCK, APMSF, AEBSEF, and aprotinin, amongst others, will inhibit Trypsin.
Serine protease inhibitors, including DFP, TLCK, APMSF, AEBSEF, and aprotinin, amongst others, will inhibit Trypsin.
Components
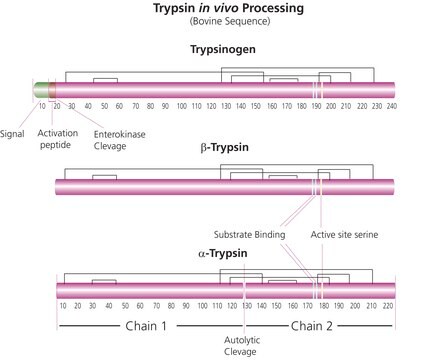
Trypsin consists of a single chain polypeptide of 223 amino acid residues, produced by the removal of the N-terminal hexapeptide from trypsinogen which is cleaved at the Lys - lle peptide bond. The sequence of amino acids is cross-linked by 6 disulfide bridges. This is the native form of trypsin, beta-trypsin. BETA-trypsin can be autolyzed, cleaving at the Lys - Ser residue, to produce alpha-trypsin. Trypsin is a member of the serine protease family.
Caution
Solutions in 1 mM HCl are stable for 1 year in aliquots and stored at -20°C. The presence of Ca2+ will also diminish the self-autolysis of trypsin and maintain its stability in solution. Trypsin will also retain most of its activity in 2.0 M urea, 2.0 M guanidine HCl, or 0.1% (w/v) SDS.
Unit Definition
One BAEE unit will produce a A253 of 0.001 per minute at pH 7.6 at 25°C using BAEE as a substrate.
Preparation Note
This product is from pancreas sourced from New Zealand. It is soluble in 1 mM HCl at 1 mg/mL.
For applications that involve EDTA, solubilizing trypsin should be done with a buffered salt solution containing no Ca2+ or Mg2+.
For applications that involve EDTA, solubilizing trypsin should be done with a buffered salt solution containing no Ca2+ or Mg2+.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Resp. Sens. 1 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
TGF-beta1 and GDF5 act synergistically to drive the differentiation of human adipose stromal cells toward nucleus pulposus-like cells
Colombier P, et al.
Stem Cells, 34(3), 653-667 (2016)
Purification of rat and mouse astrocytes by immunopanning
Foo LC
Cold Spring Harbor Protocols (2013)
Purification and culture of retinal ganglion cells from rodents
Winzeler A and Wang JT
Cold Spring Harbor Protocols (2013)
Francesca Cole et al.
Nature genetics, 46(10), 1072-1080 (2014-08-26)
The ability to examine all chromatids from a single meiosis in yeast tetrads has been indispensable for defining the mechanisms of homologous recombination initiated by DNA double-strand breaks (DSBs). Using a broadly applicable strategy for the analysis of chromatids from
Rebecca M Reese et al.
Scientific reports, 10(1), 979-979 (2020-01-24)
The transient receptor potential (TRP) superfamily of ion channels has garnered significant attention by the pharmaceutical industry. In particular, TRP channels showing high levels of expression in sensory neurons such as TRPV1, TRPA1, and TRPM8, have been considered as targets
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service