S3626
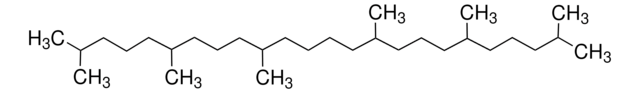
Squalene
≥98%, liquid
Synonym(s):
2,6,10,15,19,23-Hexamethyl-2,6,10,14,18,22-tetracosahexaene
About This Item
Recommended Products
Quality Level
Assay
≥98%
form
liquid
color
light yellow
refractive index
n20/D 1.494 (lit.)
bp
285 °C/25 mmHg (lit.)
mp
−75 °C (lit.)
density
0.858 g/mL at 25 °C (lit.)
application(s)
metabolomics
vitamins, nutraceuticals, and natural products
storage temp.
2-8°C
SMILES string
CC(C)=CCCC(C)=CCCC(C)=CCC\C=C(/C)CCC=C(C)CCC=C(C)C
InChI
1S/C30H50/c1-25(2)15-11-19-29(7)23-13-21-27(5)17-9-10-18-28(6)22-14-24-30(8)20-12-16-26(3)4/h15-18,23-24H,9-14,19-22H2,1-8H3/b27-17+,28-18+,29-23+,30-24+
InChI key
YYGNTYWPHWGJRM-AAJYLUCBSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- as a standard for lipid identification and quantification
- in the isolation of macrophages for parasite incubation
- as a standard for the quantification of squalene in squalene analysis of oil samples
Biochem/physiol Actions
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Asp. Tox. 1
Storage Class Code
10 - Combustible liquids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Terpenes comprise the largest and most diverse class of secondary metabolites; approximately 55,000 compounds have been identified to date.
Biosynthesis of cholesterol generally takes place in the endoplasmic reticulum of hepatic cells and begins with acetyl- CoA, which is mainly derived from an oxidation reaction in the mitochondria. Acetyl-CoA and acetoacetyl-CoA are converted to 3-hydroxy- 3-methylglutaryl-CoA (HMG-CoA) by HMG-CoA synthase.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service