G3664
Glutathione Reductase from baker′s yeast (S. cerevisiae)
ammonium sulfate suspension, 100-300 units/mg protein (biuret)
Synonym(s):
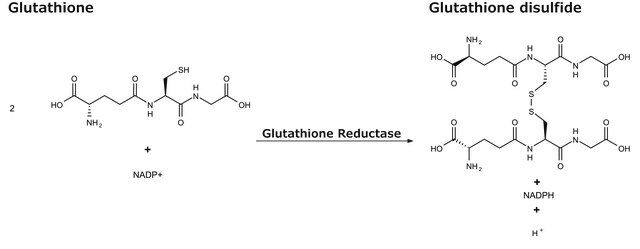
GR, NAD(P)H:oxidized-glutathione oxidoreductase
About This Item
Recommended Products
form
ammonium sulfate suspension
Quality Level
specific activity
100-300 units/mg protein (biuret)
mol wt
118 kDa
UniProt accession no.
foreign activity
G-6-PDH, 6-PGDH, and NADPH oxidase ≤0.01%
lipoamide dehydrogenase ≤0.1%
storage temp.
2-8°C
Gene Information
bakers yeast ... GLR1(856014)
Looking for similar products? Visit Product Comparison Guide
General description
Application
- in the glutathione assay to determine glutathione concentration.
- as a standard in the generation of calibration curve.
- as an antigen to measure plasma activity of GR.
- for quantifying the myocardial tissue glutathione content using a glutathione reductase-5,5′-dithiobis (2-nitrobenzoic acid)-based enzymatic recycling assay
- for the quantification of reduced glutathione (GSH) in the oocytes, using a slightly modified microglutathione assay, obtained from prepubertal gilts
- for the preparation of total GSSG (glutathione disulphide) + GSH measurement, where all available GSSG was reduced to GSH, in rat lens
- for the quantification of intracellular reduced glutathione (GSH) in the oocytes obtained from rats
Biochem/physiol Actions
Unit Definition
Physical form
Preparation Note
inhibitor
related product
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Resp. Sens. 1
Storage Class Code
12 - Non Combustible Liquids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Instructions for working with enzymes supplied as ammonium sulfate suspensions
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service