C8511
Cathepsin C from bovine spleen
Type X, lyophilized powder, ≥5 units/mg protein
Synonym(s):
Dipeptidyl aminopeptidase, Dipeptidyl peptidase I
About This Item
Recommended Products
biological source
bovine spleen
Quality Level
type
Type X
Assay
>25% protein (biuret)
form
lyophilized powder
specific activity
≥5 units/mg protein
composition
Protein, ≥25% biuret
manufacturer/tradename
Sigma-Aldrich
storage condition
OK to freeze (Unstable. Keep frozen)
concentration
≥5 unit/mg protein
technique(s)
activity assay: suitable
suitability
suitable for molecular biology
application(s)
life science and biopharma
shipped in
dry ice
storage temp.
−20°C
Gene Information
cow ... CTSC(352958)
Related Categories
General description
Dipeptidyl peptidase I (DPPI), also known as cathepsin C, is an abundant lysosomal cysteine protease from the papain superfamily with a molecular weight of approximately 200 kDa. It is widely expressed in a variety of mammalian tissues, with the highest levels found in the lungs, kidneys, liver, and spleen, and relatively lower levels in the brain.
DPPI is the only member of its family that is functional as a tetramer, consisting of four identical subunits, each composed of an N-terminal fragment, a heavy chain, and a light chain. It is identified as one of the multifaceted protease-processing machines, having been shown to function beyond its role as a non-specific lysosomal protease.
Application
Biochem/physiol Actions
Cat C participates in neutrophil recruitment and production of chemokines and cytokines in many inflammatory diseases. Cathepsin C plays a crucial role as an essential enzyme in activating granule serine proteases in cytotoxic T lymphocytes, natural killer cells (granzymes A and B), mast cells (chymase and tryptase), and neutrophils (cathepsin G, proteinase 3, and elastase).
Caution
Unit Definition
Physical form
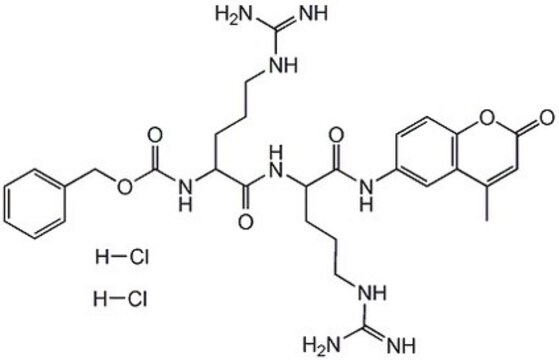
substrate
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service