07-6130
DL-Dithiothreitol
≥99.0%
Synonym(s):
threo-1,4-Dimercapto-2,3-butanediol, Cleland’s reagent, DTT
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
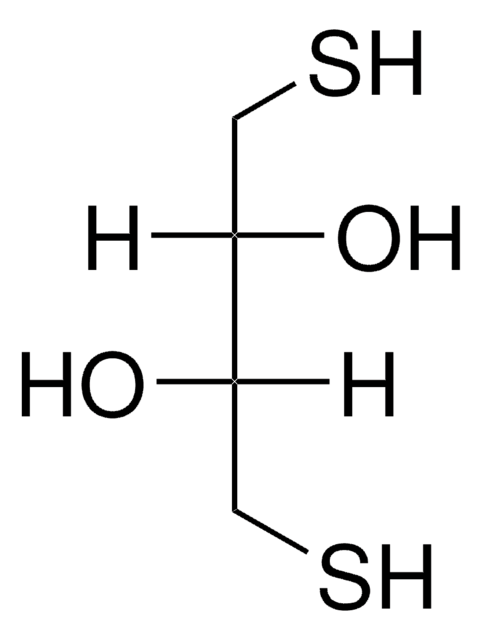
HSCH2CH(OH)CH(OH)CH2SH
CAS Number:
Molecular Weight:
154.25
Beilstein:
1719757
EC Number:
MDL number:
UNSPSC Code:
12352209
PubChem Substance ID:
Recommended Products
Assay
≥99.0%
availability
available only in Japan
mp
41-44 °C (lit.)
SMILES string
O[C@H](CS)[C@H](O)CS
InChI
1S/C4H10O2S2/c5-3(1-7)4(6)2-8/h3-8H,1-2H2/t3-,4-/m1/s1
InChI key
VHJLVAABSRFDPM-QWWZWVQMSA-N
Looking for similar products? Visit Product Comparison Guide
Application
An excellent reagent for maintaining SH groups in reduced state; quantitatively reduces disulfides. DTT is effective in sample buffers for reducing protein disulfide bonds prior to SDS-PAGE. DTT can also be used for reducing the disulfide bridge of the cross-linker N,N′-bis(acryloyl)cystamine to break apart the matrix of a polyacrylamide gel. DTT is less pungent and is less toxic than 2-mercaptoethanol. Typically, a seven fold lower concentration of DTT (100 mM) is needed than is used for 2-mercaptoethanol (5% v/v, 700 mM).
Suitability
for biological purposes
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1 - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Xueqian Liu et al.
The Journal of biological chemistry, 288(10), 7230-7240 (2013-01-25)
In Pichia pastoris, the peroxisomal targeting signal 2 (PTS2)-dependent peroxisomal matrix protein import pathway requires the receptor, Pex7, and its co-receptor Pex20. A conserved lysine (Lys(19)) near the N terminus of Pex20 is required for its polyubiquitination and proteasomal degradation
Jin-Gu Lee et al.
Nature communications, 4, 1568-1568 (2013-03-07)
In eukaryotes, deubiquitinases (DUBs) remove ubiquitin conjugates from diverse substrates, altering their stabilities, localizations or activities. Here we show that many DUBs of the USP and UCH subfamilies can be reversibly inactivated upon oxidation by reactive oxygen species in vitro
Mojtaba Abdul Roda et al.
PloS one, 9(5), e97594-e97594 (2014-05-20)
A novel neutrophil chemoattractant derived from collagen, proline-glycine-proline (PGP), has been recently characterized in chronic obstructive pulmonary disease (COPD). This peptide is derived via the proteolytic activity of matrix metalloproteases (MMP's)-8/9 and PE, enzymes produced by neutrophils and present in
Changes of metal-induced toxicity by H2O 2/NO modulators in Scenedesmus quadricauda (Chlorophyceae).
František Štork et al.
Environmental science and pollution research international, 20(8), 5502-5511 (2013-02-26)
Effect of nitric oxide donor (sodium nitroprusside, SNP, 500 μM) or hydrogen peroxide scavenger (dithiothreitol, DTT, 500 μM) on cadmium (Cd) or copper (Cu) uptake (150 μM solutions) and toxicity using Scenedesmus quadricauda was studied. Combined treatments (Cd or Cu
Hanayo Ueoka-Nakanishi et al.
The FEBS journal, 280(14), 3220-3231 (2013-04-26)
Thioredoxin (Trx) is a key player in redox homeostasis in various cells, modulating the functions of target proteins by catalyzing a thiol-disulfide exchange reaction. Target proteins of cytosolic Trx-h of higher plants were studied, particularly in the plasma membrane, because
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service