496383
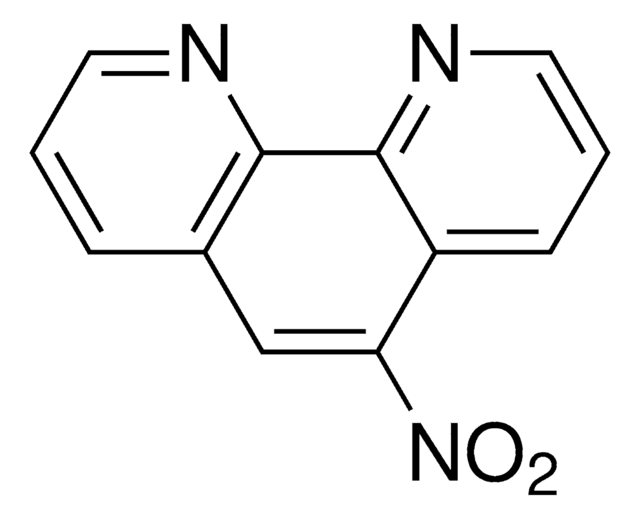
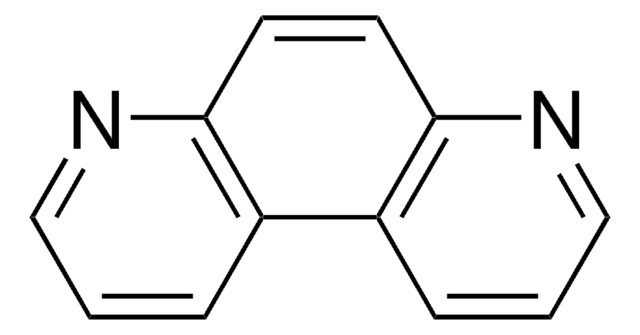
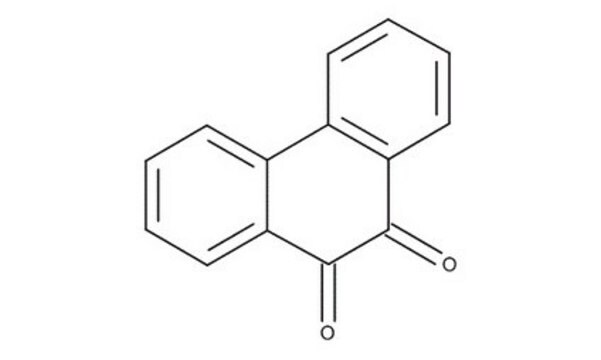
1,10-Phenanthroline-5,6-dione
97%
Synonym(s):
Stahl phd oxidant, phd
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C12H6N2O2
CAS Number:
Molecular Weight:
210.19
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
mp
260 °C (dec.) (lit.)
functional group
ketone
SMILES string
O=C1C(=O)c2cccnc2-c3ncccc13
InChI
1S/C12H6N2O2/c15-11-7-3-1-5-13-9(7)10-8(12(11)16)4-2-6-14-10/h1-6H
InChI key
KCALAFIVPCAXJI-UHFFFAOYSA-N
General description
1,10-Phenanthroline-5,6-dione (phendio) forms Cu(II) and Ag(I) phendio complexes, which show potent anti-fungal and anti-cancer activity. The modification of glassy carbon (GC) electrodes with phendio complexes of transition metals leads to the catalytic oxidation of NADH at low overpotential.
Application
1,10-Phenanthroline-5,6-dione may be used in the preparation of homo- and heterometallic complexes with early transition metal ions.
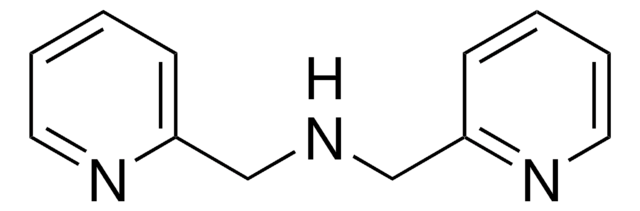
A Bifunctional quinone oxidant which, when used in conjunction with Zn2+ catalysts, is used to affect the aerobic oxidation of secondary amines to a variety of value added motifs, including indoles.
Bioinspired Aerobic Oxidation of Secondary Amines and Nitrogen Heterocycles with a Bifunctional Quinone Catalyst
Bioinspired Aerobic Oxidation of Secondary Amines and Nitrogen Heterocycles with a Bifunctional Quinone Catalyst
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Smita Ghosh et al.
Journal of inorganic biochemistry, 100(3), 331-343 (2006-01-18)
Complexes of the type [Co(LL)2Cl2]Cl, where LL = N,N'-ethylenediamine (en), 2,2'-bipyridine (bpy), 1,10-phenanthroline (phen), 1,10-phenanthroline-5,6-dione (phendione) and dipyrido[3,2-a:2',3'-c]phenazine (dppz) have been synthesized and characterized by elemental analyses, IR, UV-visible and NMR spectroscopy. Crystal structure of [Co(phendione)2Cl2]Cl x 0.5 HCl x
M V del Pozo et al.
Analytical chemistry, 77(8), 2550-2557 (2005-04-15)
A surface-based method for the study of the interactions of DNA with redox-active 1,10-phenantroline-5,6-dione (phen-dione) osmium complexes is described. The study was carried out using gold electrodes modified with DNA via adsorption and [Os(bpy)(2)(phe-dione)](3+/2+) (bpy = 2,2'-bipyridyl) or [Os(phen)(2)(phen-dione)](3+/2+) (phen
E Lorenzo et al.
Biosensors & bioelectronics, 13(3-4), 319-332 (1998-06-27)
Various strategies based on the use of chemically modified electrodes for the development of amperometric biosensors are described. Particular emphasis is placed on materials capable of catalyzing the oxidation of NADH and coupling these with enzymatic activities for biosensor construction.
1, 10-Phenanthroline-5, 6-dione as a building block for the synthesis of homo-and heterometallic complexes.
Calderazzo F, et al.
Inorgorganica Chimica Acta, 330(1), 136-142 (2002)
Yu-Min Song et al.
Journal of inorganic biochemistry, 103(3), 396-400 (2009-01-13)
Three new solid complexes have been synthesized by the reaction of rare earth(III) nitrate with the first ligand curcumin (HL) and the second ligand 1,10-phenanthroline-5,6-dione (L') in alcohol solution (pH=6.5-7.0). The composition of the complexes has been characterized by elemental
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service