115665
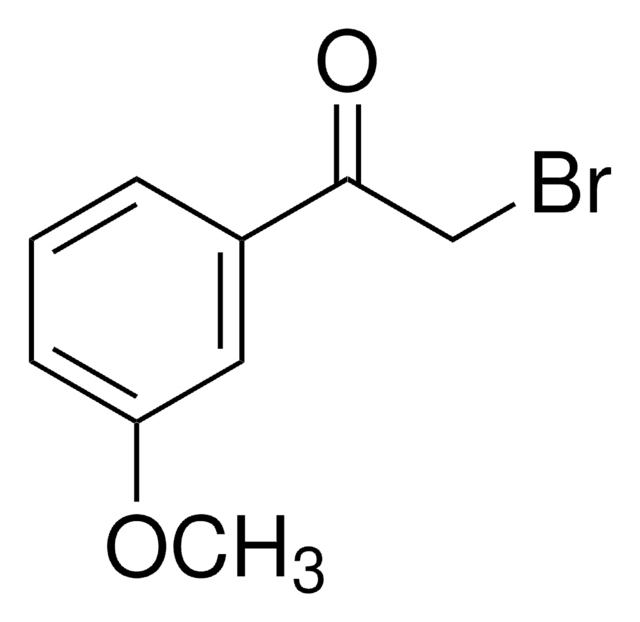
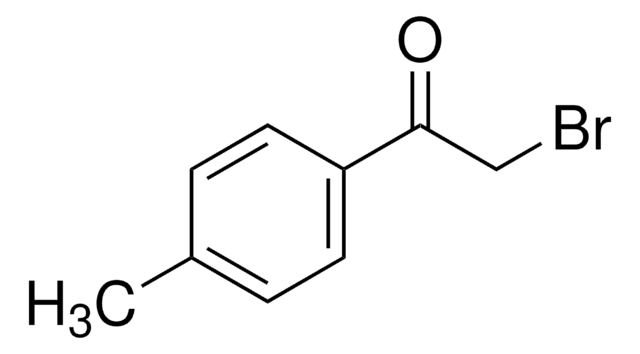
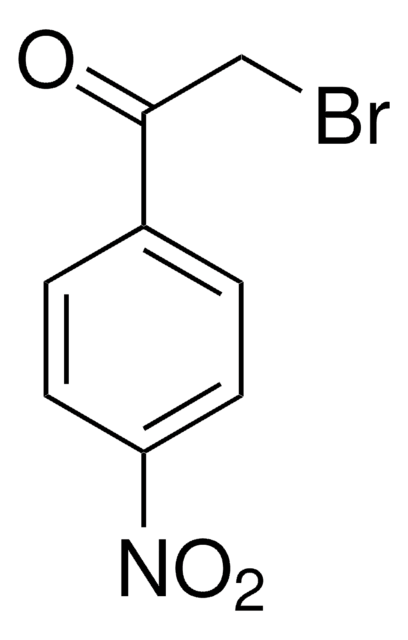
2-Bromo-4′-methoxyacetophenone
97%
Synonym(s):
ω-Bromo-4-methoxyacetophenone, 4-Methoxyphenacyl bromide
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
CH3OC6H4COCH2Br
CAS Number:
Molecular Weight:
229.07
Beilstein:
743112
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
form
solid
mp
69-71 °C (lit.)
functional group
bromo
storage temp.
2-8°C
SMILES string
COc1ccc(cc1)C(=O)CBr
InChI
1S/C9H9BrO2/c1-12-8-4-2-7(3-5-8)9(11)6-10/h2-5H,6H2,1H3
InChI key
XQJAHBHCLXUGEP-UHFFFAOYSA-N
Gene Information
human ... PTPN6(5777)
Looking for similar products? Visit Product Comparison Guide
Application
2-Bromo-4′-methoxyacetophenone has been used in the synthesis of light-responsive diblock copolymers containing para-methoxyphenacyl photocleavable side groups.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1 - Skin Corr. 1B - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Synthesis of diblock copolymers bearing p-methoxyphenacyl side groups.
Polym. Chem., 2(10), 2284-2292 (2011)
Guido J Noguera et al.
European journal of pharmaceutical sciences : official journal of the European Federation for Pharmaceutical Sciences, 78, 190-197 (2015-07-27)
A set of 4-arylthiazolylhydrazones derived from 1-indanones (TZHs) previously synthesized and assayed against Trypanosoma cruzi, the causative agent of Chagas disease, were explored in terms of conformational analysis. We found that TZHs can adopt four minimum energy conformations: cis (A
Daniel I Perez et al.
Journal of medicinal chemistry, 54(12), 4042-4056 (2011-04-20)
Development of kinase-targeted therapies for central nervous system (CNS) diseases is a great challenge. Glycogen synthase kinase 3 (GSK-3) offers a great potential for severe CNS unmet diseases, being one of the inhibitors on clinical trials for different tauopathies. Following
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service