All Photos(1)
About This Item
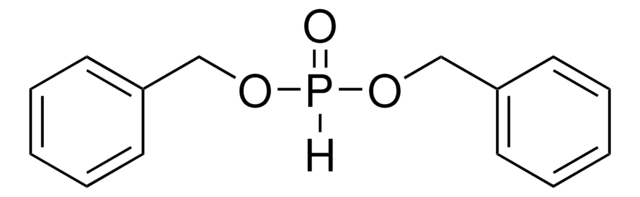
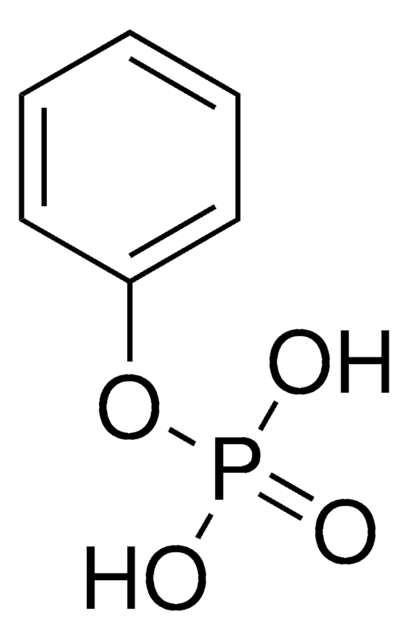
Linear Formula:
(C6H5CH2O)2P(O)(OH)
CAS Number:
Molecular Weight:
278.24
Beilstein:
2055755
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
99%
form
powder
mp
78-80 °C (lit.)
storage temp.
2-8°C
SMILES string
OP(=O)(OCc1ccccc1)OCc2ccccc2
InChI
1S/C14H15O4P/c15-19(16,17-11-13-7-3-1-4-8-13)18-12-14-9-5-2-6-10-14/h1-10H,11-12H2,(H,15,16)
InChI key
HDFFVHSMHLDSLO-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Dibenzyl phosphate (DBzP) can be used:
- To promote the monoselective ortho-C-H alkylation of N-quinolyl benzamides with primary and secondary alkyl iodides.
- For the ring-opening reaction of epoxide such as benzylglycidol to synthesize dihydroxyacetone phosphate (DHAP).
- As a reactant for the synthesis of stereospecific 1,2-trans glycosyl phosphates.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
An approach towards the synthesis of 1, 2-trans glycosyl phosphates via iodonium ion assisted activation of thioglycosides.
Veeneman GH, et al.
Tetrahedron Letters, 32(43), 6175-6178 (1991)
Lewis acid mediated regioselective ring opening of benzylglycidol with dibenzyl phosphate: short and attractive synthesis of dihydroxyacetone phosphate.
Meyer O, et al.
Organic Letters, 8(19), 4347-4350 (2006)
Syntheses of glycosyl phosphates by phase transfer catalysis.
Roy R, et al.
Canadian Journal of Chemistry, 69(9), 1462-1467 (1991)
Pd-Catalyzed Monoselective ortho-C-H Alkylation of N-Quinolyl Benzamides: Evidence for Stereoretentive Coupling of Secondary Alkyl Iodides.
Zhang SY, et al.
Journal of the American Chemical Society, 137(1), 531-539 (2014)
Biosynthetic studies on the ?-glucosidase inhibitor acarbose: the chemical synthesis of dTDP-4-amino-4, 6-dideoxy-?-d-glucose.
Bowers SG, et al.
Carbohydrate Research, 337(4), 297-304 (2002)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service