232491
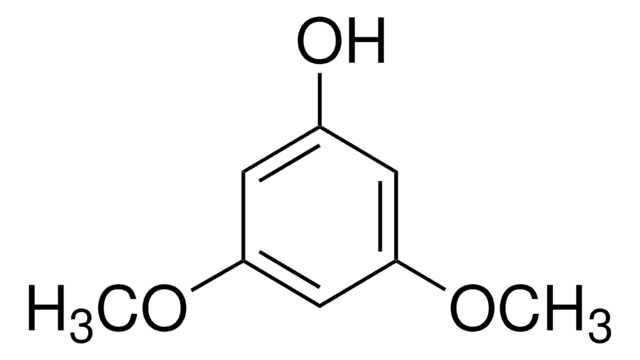
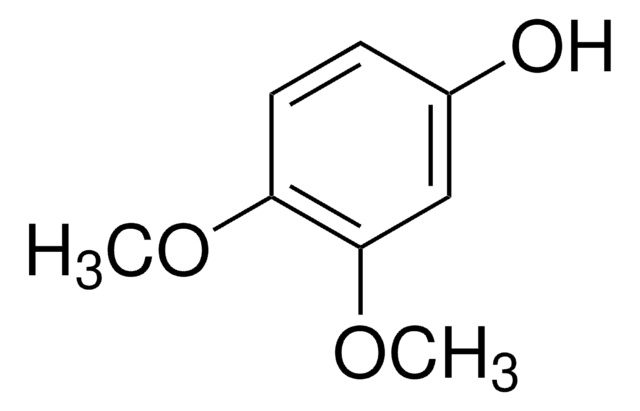
5-Methoxyresorcinol
98%
Synonym(s):
3,5-Dihydroxyanisole, Phloroglucinol monomethyl ether
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
CH3OC6H3-1,3-(OH)2
CAS Number:
Molecular Weight:
140.14
Beilstein:
1423578
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
bp
188-189 °C/12 mmHg (lit.)
mp
78-80 °C (lit.)
SMILES string
COc1cc(O)cc(O)c1
InChI
1S/C7H8O3/c1-10-7-3-5(8)2-6(9)4-7/h2-4,8-9H,1H3
InChI key
HDVRLUFGYQYLFJ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Kinetics of the aroxyl radical-scavenging action of 5-methoxyresorcinol has been investigated. The kinetics of reaction of 5,7-diisopropyl-tocopheroxyl radical (Toc) with 5-methoxyresorcinol has been measured by stopped-flow spectrophotometer.
Application
5-Methoxyresorcinol was used in synthesis of:
- isorobustin
- substituted linear and angular benzofurocoumarins
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Shuji Mitani et al.
Journal of agricultural and food chemistry, 56(12), 4406-4417 (2008-05-27)
Kinetic study of the aroxyl radical-scavenging action of catechins (epicatechin (EC), epicatechin gallate (ECG), epigallocatechin (EGC), and epigallocatechin gallate (EGCG)) and related compounds (methyl gallate (MG), 4-methylcatechol (MC), and 5-methoxyresorcinol (MR)) has been performed. The second-order rate constant ( k
Synthesis of linear and angular benzofurocoumarins.
Teran C, et al.
Synthesis, 1997(12), 1384-1386 (1997)
Kazuo Mukai et al.
Free radical biology & medicine, 38(9), 1243-1256 (2005-04-06)
The reaction rates (k(r)) of 5,7-diisopropyl-tocopheroxyl radical (Toc) with catechins (epicatechin (EC), epicatechin gallate (ECG), epigallocatechin (EGC), epigallocatechin gallate (EGCG)) and related compounds (methyl gallate (MG), 4-methylcatechol (MC), and 5-methoxyresorcinol (MR)) have been measured by stopped-flow spectrophotometer. The k(r) values
Total synthesis of isorobustin.
Barton DHR, et al.
Tetrahedron Letters, 31(51), 7449-7452 (1990)
J U Kreft et al.
European journal of biochemistry, 226(3), 945-951 (1994-12-15)
The previously studied complete methyl transfer sequence of tetrahydrofolate-dependent O-demethylation catalyzed by Holophaga foetida strain TMBS4 extracts was separated into two steps using cobalamins as non-physiological substrates: electrochemically produced cob(I) alamin served as methyl acceptor for phenyl methyl ether demethylation
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service