120804
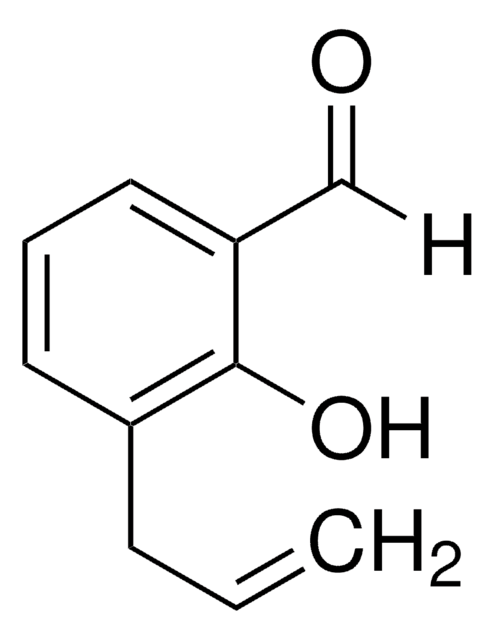
o-Vanillin
99%
Synonym(s):
2-Hydroxy-3-methoxybenzaldehyde, 2-Hydroxy-m-anisaldehyde, 3-Methoxysalicylaldehyde
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
CH3OC6H3-2-(OH)CHO
CAS Number:
Molecular Weight:
152.15
Beilstein:
471913
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
99%
form
solid
bp
265-266 °C (lit.)
mp
40-42 °C (lit.)
functional group
aldehyde
SMILES string
COc1cccc(C=O)c1O
InChI
1S/C8H8O3/c1-11-7-4-2-3-6(5-9)8(7)10/h2-5,10H,1H3
InChI key
JJVNINGBHGBWJH-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
o-Vanillin is a building block commonly used in the synthesis of schiff-base ligands.
Application
o-Vanillin has been used to study the solvent-free reaction between o-vanillin and p-toluidine using NMR, DSC and XRD analysis. It was used in the synthesis of new ligand for Fe(III) and Al(lII).
Biochem/physiol Actions
o-Vanillin induces DNA damage as detected by comet assay.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2
Storage Class Code
13 - Non Combustible Solids
WGK
WGK 3
Flash Point(F)
235.4 °F - closed cup - (External MSDS)
Flash Point(C)
113 °C - closed cup - (External MSDS)
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
G Frenzilli et al.
Mutation research, 468(2), 93-108 (2000-07-07)
To validate the alkaline single cell gel (SCG) assay as a tool for the detection of DNA damage in human leukocytes, we investigated the in vitro activity of 18 chemicals. Thirteen of these chemicals (pyrene (PY), benzo(a)pyrene (BaP), cyclophosphamide (CP)
Peter Temitope Adeboye et al.
Scientific reports, 7, 42635-42635 (2017-02-17)
The ability of Saccharomyces cerevisiae to catabolize phenolic compounds remains to be fully elucidated. Conversion of coniferyl aldehyde, ferulic acid and p-coumaric acid by S. cerevisiae under aerobic conditions was previously reported. A conversion pathway was also proposed. In the
Li-Jun Ru et al.
Molecules (Basel, Switzerland), 23(7) (2018-07-14)
A self-assembled ZnII-NdIII heterohexanuclear coordination compound [Zn₄Nd₂(L)₄(bdc)₂]·2NO₃ based on a hexadentate Salamo-like chelating ligand (H₂L = 1,2-bis(3-methoxysalicylideneaminooxy)ethane]) and H₂bdc (H₂bdc = terephthalic acid) has been synthesized and characterized by elemental analyses, IR and UV/Vis spectra, and X-ray crystallography. Two crystallographically
Mechanically induced reactions in organic solids: liquid eutectics or solid-state processes?
Dolotko O, et al.
New. J. Chem., 34(1), 25-28 (2010)
Afzal Hussain et al.
Scientific reports, 9(1), 5237-5237 (2019-03-29)
Copper-based compounds are promising entities for target-specific next-generation anticancer and NSAIDS therapeutics. In lieu of this, benzimidazole scaffold plays an important role, because of their wide variety of potential functionalizations and coordination modes. Herein, we report three copper complexes 1-3
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 120804-500G | 4061838716538 |
| 120804-100G | 4061838716477 |
| 120804-10G | 4061838716491 |
| 120804-10KG |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service