M2272
Melittin from honey bee venom
≥85% (HPLC)
About This Item
Produits recommandés
Source biologique
animal (Apis mellifera)
Niveau de qualité
Pureté
≥85% (HPLC)
Forme
lyophilized powder
Poids mol.
~_2.8 kDa
Solubilité
water: 5.00-5.20 mg/mL, clear, colorless to faintly yellow
Spectre d'activité de l'antibiotique
Gram-negative bacteria
Gram-positive bacteria
Activité étrangère
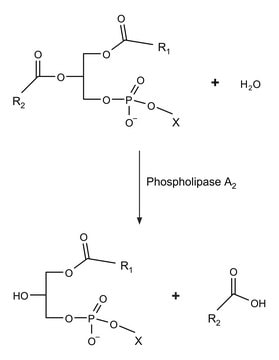
Phospholipase A2 ≤0.5%
Mode d’action
cell membrane | interferes
Température de stockage
−20°C
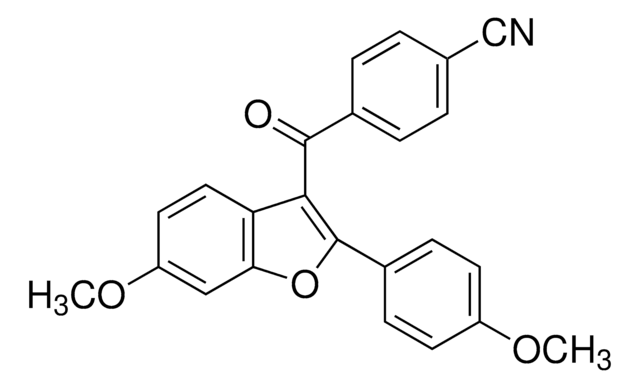
Chaîne SMILES
CC[C@H](C)[C@H](NC(=O)CN)C(=O)NCC(=O)N[C@@H](C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)N[C@@H](CC(C)C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CO)C(=O)N[C@@H](Cc2c[nH]c3ccccc23)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(N)=O
InChI
1S/C131H229N39O31/c1-23-71(16)102(163-97(176)60-135)122(194)146-62-98(177)148-74(19)109(181)164-100(69(12)13)124(196)160-88(55-65(4)5)116(188)155-84(41-30-33-51-134)115(187)165-101(70(14)15)125(197)161-90(57-67(8)9)118(190)168-106(77(22)173)128(200)169-105(76(21)172)123(195)147-63-99(178)150-92(58-68(10)11)129(201)170-54-36-44-94(170)121(193)149-75(20)108(180)158-89(56-66(6)7)117(189)166-104(73(18)25-3)127(199)162-93(64-171)120(192)159-91(59-78-61-145-80-38-27-26-37-79(78)80)119(191)167-103(72(17)24-2)126(198)157-83(40-29-32-50-133)111(183)154-85(42-34-52-143-130(139)140)112(184)152-82(39-28-31-49-132)110(182)153-86(43-35-53-144-131(141)142)113(185)156-87(46-48-96(137)175)114(186)151-81(107(138)179)45-47-95(136)174/h26-27,37-38,61,65-77,81-94,100-106,145,171-173H,23-25,28-36,39-60,62-64,132-135H2,1-22H3,(H2,136,174)(H2,137,175)(H2,138,179)(H,146,194)(H,147,195)(H,148,177)(H,149,193)(H,150,178)(H,151,186)(H,152,184)(H,153,182)(H,154,183)(H,155,188)(H,156,185)(H,157,198)(H,158,180)(H,159,192)(H,160,196)(H,161,197)(H,162,199)(H,163,176)(H,164,181)(H,165,187)(H,166,189)(H,167,191)(H,168,190)(H,169,200)(H4,139,140,143)(H4,141,142,144)/t71-,72-,73-,74-,75-,76+,77+,81-,82-,83-,84-,85-,86-,87-,88-,89-,90-,91-,92-,93-,94-,100-,101-,102-,103-,104-,105-,106-/m0/s1
Clé InChI
VDXZNPDIRNWWCW-JFTDCZMZSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
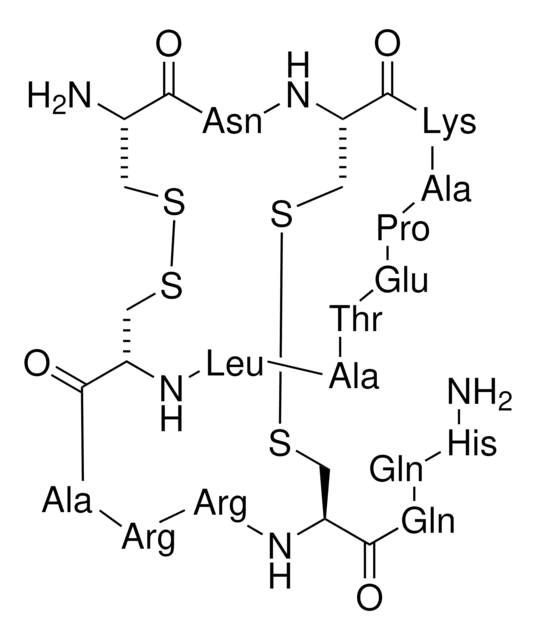
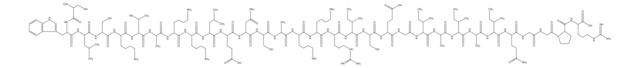
Amino Acid Sequence
Description générale
Application
- In 3-(4, 5-dimethyl thiazol-2-yl)-2,5diphenyl tetrazolium bromide (MTT) assay to determine its cytotoxicity effect on the growth of human cell lines.
- To study the anti-microbial activity of melittin on the growth of Borrelia burgdorferi in in vitro conditions.
- As a positive control in hemolysis assay and as a cytotoxic agent against HeLa cells.
Actions biochimiques/physiologiques
Autres remarques
Mention d'avertissement
Danger
Mentions de danger
Conseils de prudence
Classification des risques
Acute Tox. 3 Dermal - Acute Tox. 3 Oral
Code de la classe de stockage
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Bee Venom (Apis cerana indica) to several human cell lines: HeLa,
WiDr and Vero.
Articles
With bacterial resistance and emerging infectious diseases becoming potential threats to humans, ribosomally synthesized antimicrobial peptides have become a promising focus area in antibiotic research.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique