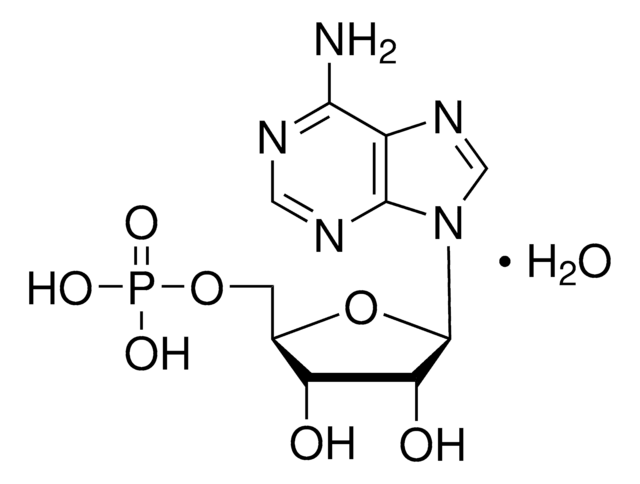
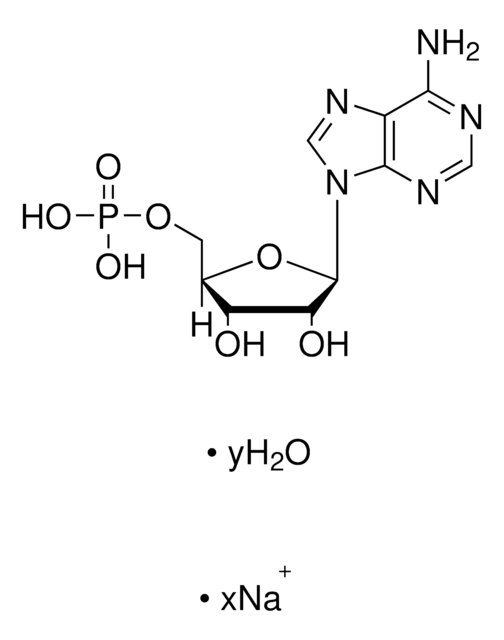
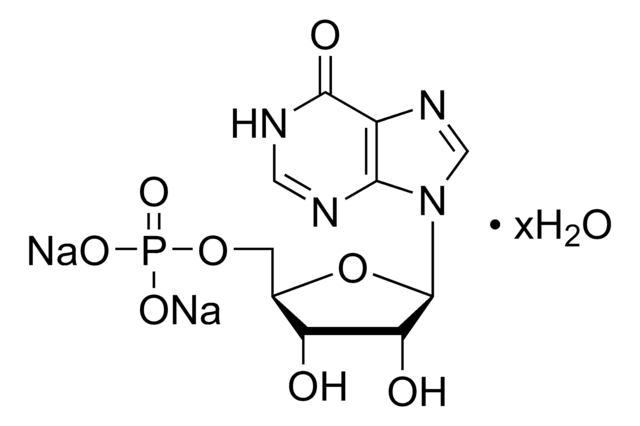
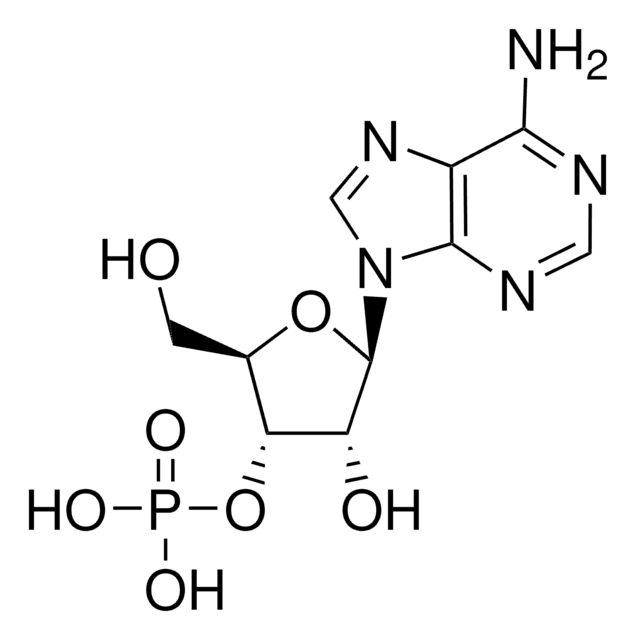
A1271
Adenosine 5′-monophosphate–Agarose
lyophilized powder
Se connecterpour consulter vos tarifs contractuels et ceux de votre entreprise/organisme
About This Item
Numéro MDL:
Code UNSPSC :
41106500
eCl@ss :
32160414
ID de substance PubChem :
Nomenclature NACRES :
NA.56
Produits recommandés
Source biologique
plant (Sea weed)
Forme
lyophilized powder
Ampleur du marquage
1-5 μmol per mL
Matrice
cross-linked 4% beaded agarose
Activation de la matrice
cyanogen bromide
Fixation de matrice
C-8
Espaceur de matrice
9 atoms
Température de stockage
−20°C
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Catégories apparentées
Application
Adenosine 5′-monophosphate Agarose (5′-AMP agarose) has been used in affinity chromatography to isolate β and gamma glutamate decarboxylase, which is important for controlling gamma-aminobutyric acid (GABA) synthesis in brain.
Forme physique
Lyophilized powder stabilized with lactose
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
Eyeshields, Gloves, type N95 (US)
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
F James et al.
The Journal of biological chemistry, 270(38), 22344-22350 (1995-09-22)
The plant enzyme S-adenosylmethionine:methionine S-methyltransferase (EC 2.1.1.12, MMT) catalyzes the synthesis of S-methylmethionine. MMT was purified 620-fold to apparent homogeneity from leaves of Wollastonia biflora. The four-step purification included fractionation with polyethylene glycol, affinity chromatography on adenosine-agarose, anion exchange chromatography
C D Murphy et al.
Applied and environmental microbiology, 67(10), 4919-4921 (2001-09-26)
Streptomyces cattleya is unusual in that it produces fluoroacetate and 4-fluorothreonine as secondary metabolites. We now report the isolation of an NAD(+)-dependent fluoroacetaldehyde dehydrogenase from S. cattleya that mediates the oxidation of fluoroacetaldehyde to fluoroacetate. This is the first enzyme
S J Wu et al.
Journal of neurochemistry, 42(6), 1607-1612 (1984-06-01)
The interactions of two forms of porcine brain glutamate decarboxylase (beta-GAD and gamma-GAD) with the effector ATP were studied by affinity chromatography. A third form, alpha-GAD, was only slightly retarded by the affinity matrix and was eluted in the buffer
M Kato et al.
Plant physiology, 120(2), 579-586 (1999-06-11)
Caffeine synthase (CS), the S-adenosylmethionine-dependent N-methyltransferase involved in the last two steps of caffeine biosynthesis, was extracted from young tea (Camellia sinensis) leaves; the CS was purified 520-fold to apparent homogeneity and a final specific activity of 5.7 nkat mg-1
D L Martin et al.
Journal of neurochemistry, 55(2), 524-532 (1990-08-01)
A major regulatory feature of brain glutamate decarboxylase (GAD) is a cyclic reaction that controls the relative amounts of holoenzyme and apoenzyme [active and inactive GAD with and without bound pyridoxal 5'-phosphate (pyridoxal-P, the cofactor), respectively]. Previous studies have indicated
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique