BCR159
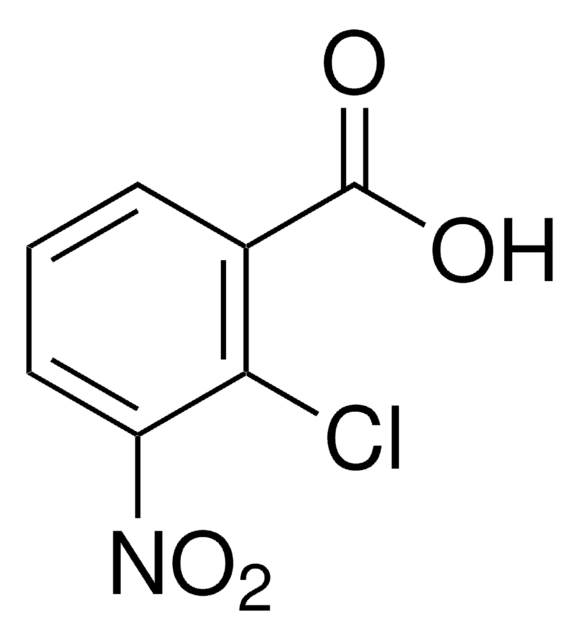
Dibenzo[a,h]pyrene
BCR®, certified reference material
About This Item
Produits recommandés
Qualité
certified reference material
Agence
BCR®
Fabricant/nom de marque
JRC
Technique(s)
HPLC: suitable
gas chromatography (GC): suitable
Format
neat
Température de stockage
2-8°C
Chaîne SMILES
c1ccc2c(c1)cc3ccc4c5ccccc5cc6ccc2c3c46
InChI
1S/C24H14/c1-3-7-19-15(5-1)13-17-9-12-22-20-8-4-2-6-16(20)14-18-10-11-21(19)23(17)24(18)22/h1-14H
Clé InChI
RXUSYFJGDZFVND-UHFFFAOYSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Remarque sur l'analyse
BCR159
Informations légales
Mention d'avertissement
Danger
Mentions de danger
Conseils de prudence
Classification des risques
Carc. 1B - Muta. 2
Code de la classe de stockage
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Listes réglementaires
Les listes réglementaires sont principalement fournies pour les produits chimiques. Seules des informations limitées peuvent être fournies ici pour les produits non chimiques. L'absence d'indication signifie qu'aucun des composants n'est répertorié. Il incombe à l'utilisateur de s'assurer de l'utilisation sûre et légale du produit.
EU REACH Annex XVII (Restriction List)
Faites votre choix parmi les versions les plus récentes :
Certificats d'analyse (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documents section.
Si vous avez besoin d'assistance, veuillez contacter Service Clients
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique![Dibenzo[a,l]pyrene BCR®, certified reference material](/deepweb/assets/sigmaaldrich/product/structures/937/930/3e2321b0-d54a-46c2-bb84-007bb57eb381/640/3e2321b0-d54a-46c2-bb84-007bb57eb381.png)
![Dibenzo[a,e]pyrene BCR®, certified reference material](/deepweb/assets/sigmaaldrich/product/structures/276/556/dd214831-9057-438a-b33d-aa10f7b4e48c/640/dd214831-9057-438a-b33d-aa10f7b4e48c.png)
![Benzo[a]pyrène ≥96% (HPLC)](/deepweb/assets/sigmaaldrich/product/structures/253/820/be96d879-1811-46c0-8f11-612019691c2d/640/be96d879-1811-46c0-8f11-612019691c2d.png)