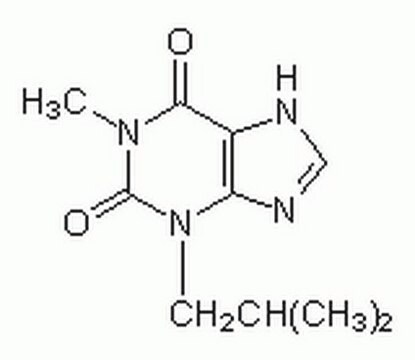
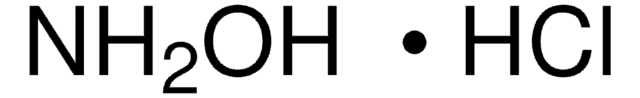
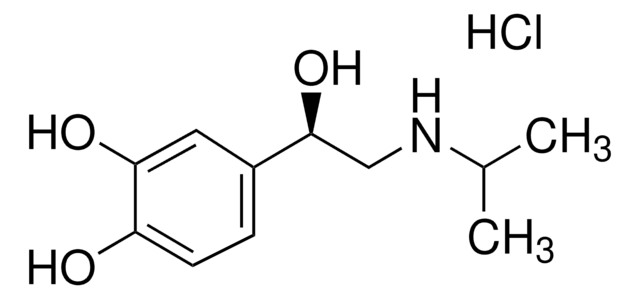
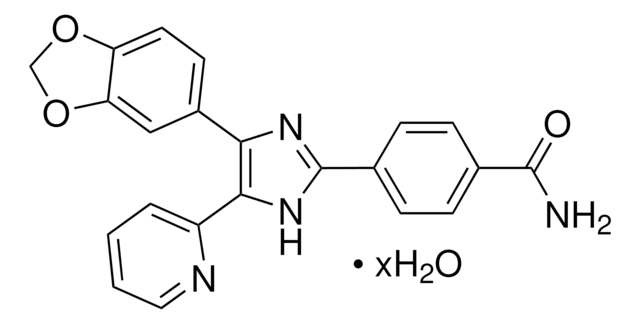
344282
Forskolin
from Coleus forskohlii, ≥97% (HPLC), liquid, positive inotropic agent, Calbiochem
Synonyme(s) :
Forskolin, Coleus forskohlii in DMSO
About This Item
Produits recommandés
Nom du produit
Forskolin, Coleus forskohlii in DMSO, The major cell-permeable diterpene isolated from the Indian plant Coleus forskohlii.
Niveau de qualité
Essai
≥97% (HPLC)
Forme
liquid
Fabricant/nom de marque
Calbiochem®
Conditions de stockage
OK to freeze
desiccated (hygroscopic)
protect from light
Conditions d'expédition
wet ice
Température de stockage
−20°C
Description générale
Conditionnement
Avertissement
Forme physique
Reconstitution
Autres remarques
Noveen, A., et al. 1996. Biochem. Biophys. Res. Commun.219, 180.
Galli, C., et al. 1995. J. Neurosci.15, 1172.
Li, X., et al. 1995. Am. J. Physiol.269, C986.
Lomo, J., et al. 1995. J. Immunol.154, 1634.
Uneyama, H., et al. 1993. J. Biol. Chem.268, 168.
Laurenza, A., et al. 1989. Trends Pharmacol. Sci.10, 442.
Adashi, E.Y., and Resnick, C.E. 1986. J. Cell. Biochem.31, 217.
Seamon, K.B., and Daly, J.W. 1986. Adv. Cyclic Nucleotide Protein Phosphorylation Res.20, 1.
Huang, R., et al. 1982. Cyclic Nucleotide Res.8, 385.
Metzger, H., and Lindner, E. 1981. IRCS Med. Sci. Biochem. Cardiovasc. System Pharmacol.9, 99.
Informations légales
Code de la classe de stockage
10 - Combustible liquids
Classe de danger pour l'eau (WGK)
WGK 2
Point d'éclair (°F)
188.6 °F - (refers to pure substance)
Point d'éclair (°C)
87 °C - (refers to pure substance)
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique